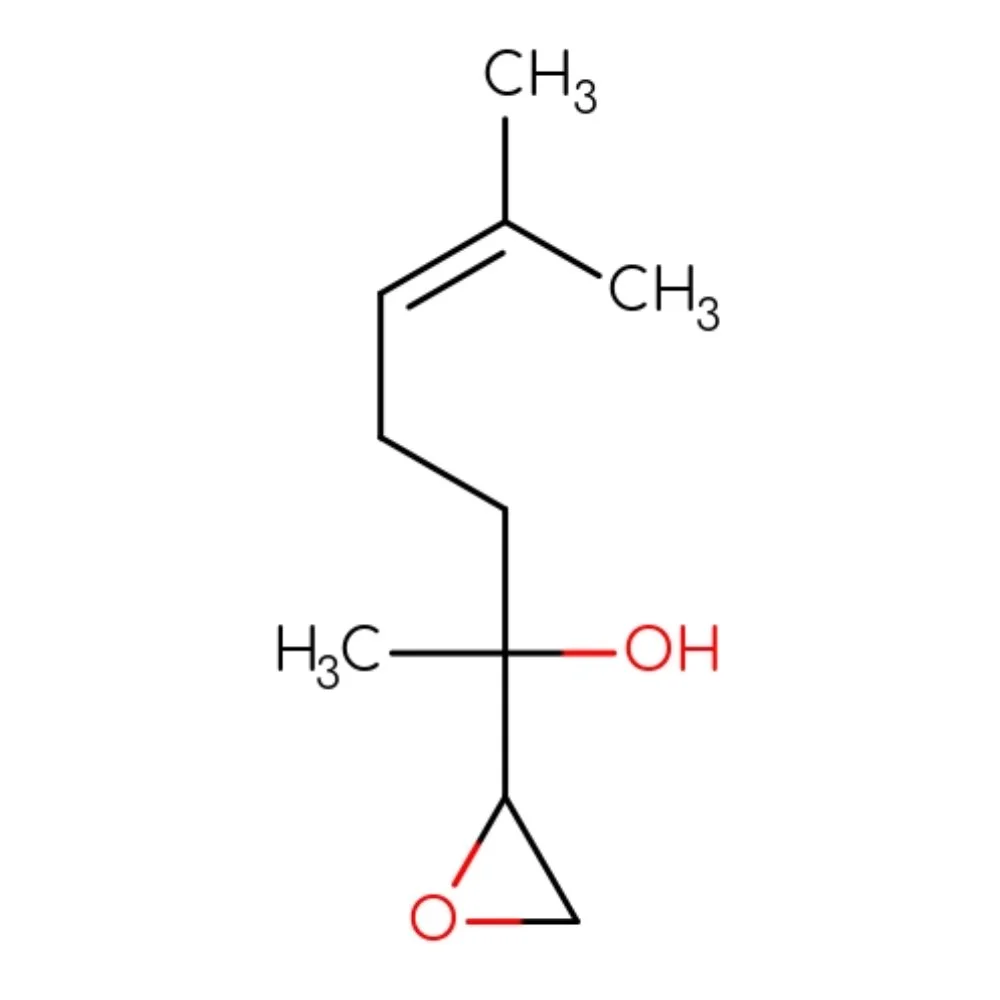
Linalool Oxide (CAS 1365-19-1) – Technical Ingredient Overview
🔎 Chemical Name — 2-Methyl-2-vinyl-5-(1-hydroxy-1-methylethyl)tetrahydrofuran
🧪 Synonyms — Linalol Oxide, Epoxylinalool, Epoxydihydrolinalool, Linalool Oxide Furanoid, 5-Ethenyltetrahydro-α,α,5-trimethyl-2-furanmethanol, α,α,5-Trimethyl-5-vinyltetrahydrofurfuryl alcohol
📂 CAS Number — 1365-19-1 (furanoid form); 60047-17-8 (mixture)
📘 FEMA Number — 3746
🔢 EC Number — 215-723-9
⚖️ Molecular Weight — 170.25 g/mol
🔬 Molecular Formula — C₁₀H₁₈O₂
📝 Odor Type — Floral
📈 Odor Strength — Medium (detection threshold: 320 ppb / 0.000032%)
👃🏼 Odor Profile — Fresh, sweet, floral, herbal, earthy, green, piney, with black tea and subtle fruity nuances
⚗️ Uses — Fine fragrance, essential oil reconstitutions, functional perfumery, flavoring agent
🧴 Appearance — Colorless to pale yellow liquid
Additional Physical Properties
🌡️ Melting Point — Data not available
🔥 Boiling Point — 188 °C (370.4 °F)
💧 Density — 0.941
🔍 Refractive Index — Data not available
💧 Solubility — Soluble in ethanol; limited water solubility
🔄 Specific Rotation — Data not available (mixture of cis/trans isomers)
✅ Purity — Typically ≥95% (commercial grade)
🧬 Biogenic Carbon Content — Available from natural sources (e.g., Eucalyptus EO extraction); synthetic grade widely produced
What is Linalool Oxide?
Linalool Oxide is a naturally occurring monoterpene oxide belonging to the oxolane (tetrahydrofuran) family. It is formed by the oxidation of linalool, one of the most abundant and ubiquitous terpene alcohols in nature. Linalool Oxide is found widely in the plant kingdom — it has been identified in Camellia sinensis (tea), Pulicaria arabica, Carica papaya, eucalyptus, and many other aromatic plants. In nature, it exists as a mixture of cis- and trans-furanoid isomers, both sharing a closely related olfactory profile.
A second structural form of linalool oxide exists based on a six-membered pyranoid ring (CAS 14049-11-7, FEMA 4593), but the furanoid (five-membered ring) form covered here is by far the most common and commercially important in perfumery and flavors.
Historical Background
Linalool Oxide's history is closely intertwined with that of linalool itself, one of the first terpene alcohols to be isolated and characterized in the 19th century. The oxide derivative was identified as a natural component of essential oils during the mid-20th century analytical revolution, when gas chromatography enabled the detailed profiling of complex plant volatiles.
Its significance grew as flavor and fragrance chemists recognized its distinctive contribution to black tea aroma — Linalool Oxide is one of the key volatiles generated during the enzymatic oxidation of tea leaves, giving black tea its characteristic sweet, floral-earthy scent. Its synthesis from linalool by controlled oxidation was developed industrially, making it broadly accessible to perfumers and flavorists. Today, it is produced both synthetically (dominant) and by extraction from natural sources such as Eucalyptus essential oil.
Olfactory Profile
Scent Family
Floral — Herbal Agrestic, with earthy, green, and fresh facets
Main Descriptors
Linalool Oxide presents a fresh, sweet floral character that is distinctly different from linalool itself. The scent profile includes:
Floral — A soft, transparent florality reminiscent of lavender and muguet, with a lifted airy quality
Black tea — A very characteristic tea-like note that dominates the profile, evocative of freshly brewed black tea leaves
Herbal / Piney — Green-herbal and coniferous facets add naturalness
Earthy — A subtle earthy, slightly dusty quality
Fruity — Delicate nuances of apple, pear, cloudberry, and lychee in the background
Sweet — An underlying powdery sweetness that ties the profile together
Intensity
Medium odor strength. Detection threshold is approximately 320 ppb (0.000032%), making it a moderately perceptible ingredient. It is not overpowering but provides noticeable lift and character to compositions.
Tenacity
Moderate tenacity. On a smelling strip, it persists for approximately 5 hours at full concentration. Substantivity on skin (both damp and dry) is rated as poor, meaning it is best used for its diffusive and blending qualities rather than as a long-lasting fixative.
Volatility
Head/Heart note. Linalool Oxide sits at the transition between top and heart, providing early freshness and lift that carries into the body of a fragrance. Vapor pressure at 20 °C is 0.0133 hPa.
Applications in Fine Fragrance
Linalool Oxide is prized for its ability to provide lift, transparency, and naturalness to a wide range of accords. It adapts well to different creative visions:
Floral accords (1–5%) — Provides additional lift and freshness to floral hearts; particularly effective in lifting rose, jasmine, and muguet compositions
Chypre accords (1–3%) — Blends harmoniously with patchouli, bridging the earthy-mossy base with brighter top notes
Tea accords (2–5%) — Essential for realistic black tea reconstitutions; its dominant tea-like character is unmatched
Fruity-floral accords (1–3%) — Enhances apple and pear facets; adds a fresh acidic fruit dimension
Essential oil reconstitutions (variable) — Invaluable in reconstructing natural essential oils, particularly lavender, eucalyptus, and tea-derived profiles
Fougère / Aromatic accords (1–2%) — Adds herbal freshness and naturalness to aromatic compositions
Performance in Formula
Fixative Power — Low; not a fixative, but contributes to the overall diffusion and blending of the composition
Radiance Effect — Strong; provides notable lift and projection to floral and herbal accords, helping them radiate in the top-heart transition
Stability — Good general stability in fragrance formulations; standard handling precautions apply
Compatibility — Excellent; blends well with a very wide range of materials including patchouli, linalool, citrus oils, floral absolutes, and woody bases
Solubility — Easily soluble in ethanol and common fragrance solvents; limited water solubility
Industrial & Technical Uses
Functional Perfumery — Used in soaps, detergents, and fabric softeners for its fresh, clean floral-herbal character and moderate burning effectiveness
Personal Care — Employed in body mists, hair care products, and cosmetic formulations for its lightweight, non-aggressive floral profile
Flavor Industry — FEMA GRAS (3746); used up to 50 ppm in flavor concentrates. Contributes floral and tea-like notes to beverage and food flavors. FDA-approved as a direct food additive under 21 CFR 172.515. JECFA Flavor Number: 1454
Cosmetics — Incorporated in home fragrance and personal care at typical perfumery use levels (1–5%)
Regulatory & Safety Overview
IFRA Status
No restriction — Linalool Oxide is not listed among IFRA restricted or prohibited materials. IFRA conformity certificates confirm it does not contain any substances prohibited by IFRA standards.
EU Cosmetics Regulation
Permitted for use; not listed among the 26 declarable allergens under EU Cosmetics Regulation (EC) No 1223/2009.
EU REACH
Registered. Does not meet the PBT/vPvB criteria of REACH, Annex XIII.
FEMA Status
GRAS (Generally Recognized As Safe) — FEMA Number 3746. GRAS Publication Numbers: 14, 25.
GHS Classification
Signal Word: Warning
Hazard Statements: H227 (Combustible liquid), H302 (Harmful if swallowed), H315 (Causes skin irritation), H319 (Causes serious eye irritation)
GHS Pictogram: GHS07 (Exclamation mark)
Precautionary Statements: P264, P270, P280, P301+P312, P305+P351+P338, P337+P313, P501
Toxicology
Sensitization — Low concern. The skin sensitization endpoint was evaluated using the Dermal Sensitization Threshold (DST) for non-reactive materials (900 μg/cm²); exposure is below the DST. Not listed as a declarable allergen.
Phototoxicity — No phototoxicity or photoallergenicity concern. Evaluated based on UV/visible spectral analysis — does not absorb significantly in the phototoxic range.
General — Not genotoxic based on available data (negative results in multiple bacterial reverse mutation assays with and without metabolic activation). A 13-week subchronic toxicity study in rats (2024) showed adverse effects only at the highest dose (800 mg/kg/day): abnormal gait, decreased locomotor activity, reduced body weight gain, and potential liver effects (increased γ-GTP, altered serum lipids). NOAEL established at 80 mg/kg/day for systemic effects. Repeated dose, reproductive, and local respiratory toxicity endpoints evaluated via TTC for Cramer Class III material — exposure levels are well below the TTC.
Environmental Considerations
Biodegradability — Readily biodegradable based on modeling (EPI Suite BIOWIN). Does not meet criteria for environmental persistence.
Bioaccumulation — Low bioaccumulative potential. Predicted fish BCF well below 2000 L/kg threshold. Log P = 1.7 (low).
Sustainability Credentials
Available from natural renewable sources (extraction from Eucalyptus and other essential oils). Natural-grade Linalool Oxide can support naturality claims. Synthetic production from linalool is efficient and well-established. No specific certifications (e.g., TÜV SÜD, ASTM D6866) widely reported for this ingredient at present.
Related Ingredients on Scentspiracy
Floral / Herbal Family:
Complementary Materials:
Patchouli Oil — Classic synergy in chypre accords; Linalool Oxide bridges the earthy base with brighter floral top notes
Hedione — Combined for transparent, radiant floral hearts with enhanced diffusion
Dihydromyrcenol — Paired for fresh, airy aromatic-floral effects in modern fougères
References
PubChem — CID 22310: Linalool, oxide (National Library of Medicine)
RIFM Fragrance Ingredient Safety Assessment, Linalool Oxide, CAS 1365-19-1 (Elsevier / RIFM)
Givaudan Fragrance Ingredients — Linalool Oxide product page
The Good Scents Company — Linalool oxide data sheet (rw1014332)
Perfumer & Flavorist (March 2013) — "Linalool Oxide" by John Wright
Safety Data Sheets: Perfumer's Apprentice (FPD-2015A-2226), Synerzine, Aurochemicals
FDA Substances Added to Food (formerly EAFUS) — Linalool Oxide entry
Yamahara et al. (2024) — "A 13-week subchronic toxicity study of linalool oxide in Crl:CD(SD) rats" (PubMed / PMC)