Indoflor (CAS 18096-62-3) Technical Ingredient Overview
🏭 Manufacturer — Symrise (Indoflor® Cryst.), IFF (Indolarome)
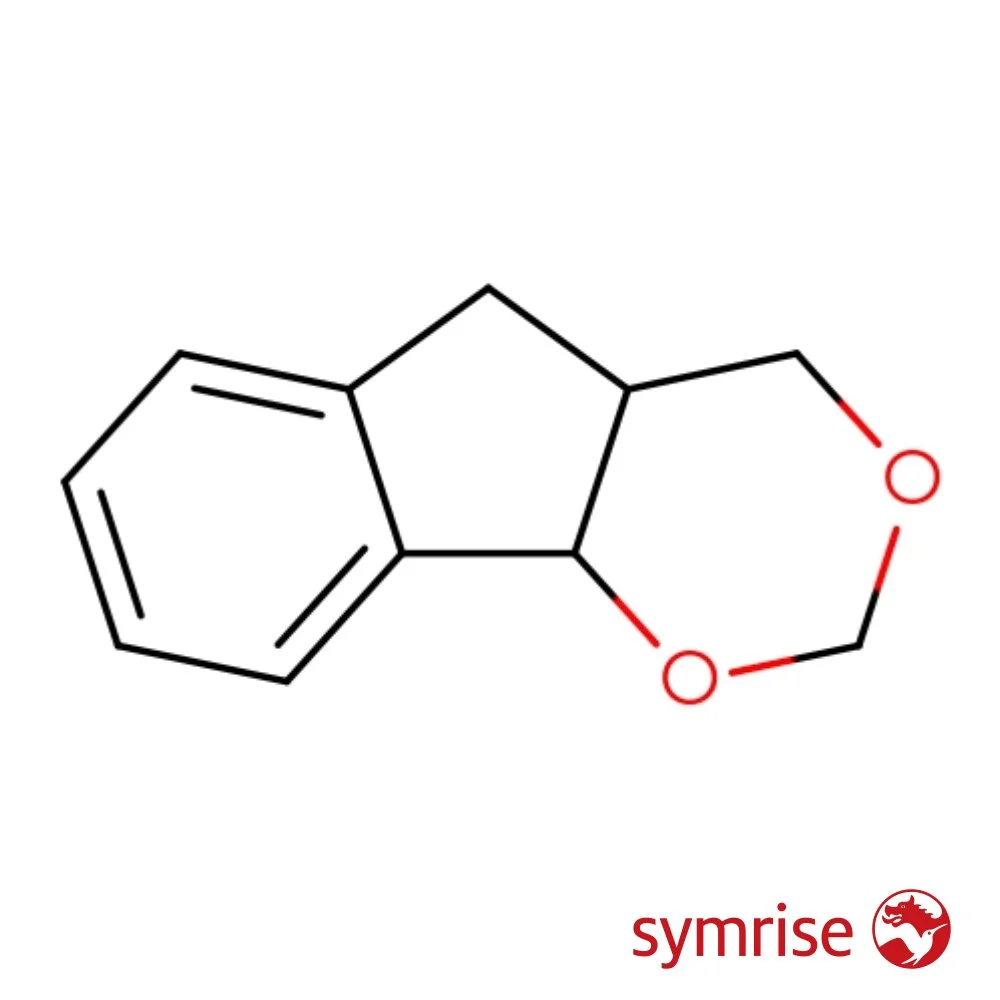
🔎 Chemical Name — 4,4a,5,9b-Tetrahydroindeno[1,2-d][1,3]dioxine
🧪 Synonyms — Indolarome, Indoletal, Florindal, Indolal, Dihydroindenyl-2,4-dioxane, Tetrahydroindenodioxin
📂 CAS Number — 18096-62-3
📘 FEMA Number — N/A
🔢 EC Number — 241-997-4
⚖️ Molecular Weight — 176.21 g/mol
🔬 Molecular Formula — C₁₁H₁₂O₂
📝 Odor Type — Animalic
📈 Odor Strength — High (recommend smelling in a 1.00% solution or less)
👃🏼 Odor Profile — Oily, animalic and civet-like, reminiscent of skatole, with indolic, earthy, jasmine and leather nuances
⚗️ Uses — Fine fragrance, functional perfumery (soaps, detergents, personal care), white flower accords
🧴 Appearance — Pale white to white crystals or agglutinant powder
Additional Physical Properties
🌡️ Melting Point — 35–36 °C @ 760 mmHg
🔥 Boiling Point — 264–283 °C @ 760 mmHg (est.)
💧 Density — 1.154 ± 0.06 g/cm³ (est.); 1.27 g/cm³ (PCW)
🔍 Refractive Index — 1.554 ± 0.02 (est.)
💧 Solubility — Soluble in alcohol; practically insoluble in water (~1,549 mg/L @ 25 °C est.)
🔄 Specific Rotation — N/A
✅ Purity — min. 99% (GC, Symrise specification)
🧬 Biogenic Carbon Content — Synthetic origin; not occurring in nature
What is Indoflor?
Indoflor is a synthetic aroma chemical belonging to the acetal chemical class. Its IUPAC name is 4,4a,5,9b-tetrahydroindeno[1,2-d][1,3]dioxine. It is an indane-dioxane derivative prepared via a Prins reaction from indene and formaldehyde in the presence of dilute sulfuric acid. The result is a crystalline solid with a powerful animalic-indolic scent profile.
Indoflor functions as a less discoloring alternative to skatole and indole, providing similar animalic and fecal-floral effects without the significant browning that natural indole or skatole can cause in finished products. This makes it highly valued in both fine fragrance and functional perfumery applications where stability and color are important considerations.
Historical Background
Indoflor was developed as part of the exploration of indane-based acetals in the mid-20th century, primarily by Haarmann & Reimer (now Symrise). The Prins reaction—a well-known acid-catalyzed condensation of an olefin with formaldehyde—was applied to indene to yield this cyclic acetal. The compound quickly gained recognition as a versatile building block for animalic and white floral accords.
Over the decades, Indoflor has been marketed under several trade names: Indoflor® Cryst. by Symrise, Indolarome by IFF, and Indoletal by various suppliers. Its ability to evoke the rich, animalic facets of natural indole—while being more stable, less volatile, and less prone to discoloration—secured its place in the modern perfumer's palette.
Olfactory Profile
Scent Family
Animalic–Floral with civet, leather, and earthy facets.
Main Descriptors
At low concentrations (≤1% in DPG), Indoflor reveals a complex scent: oily, animalic and civet-like, strongly reminiscent of skatole, with indolic, earthy, jasmine and leather nuances. At higher doses it becomes distinctly fecal and indolic; at trace levels it lends a warm, living quality to floral compositions, adding naturalness and body.
Symrise describes it as "animalic, floral, civet, leather" while Moellhausen notes "floreal, indole-like" characteristics. This duality—between raw animality and floral beauty—is what makes the ingredient so compelling.
Intensity
High. The detection threshold in air is extremely low at approximately 0.0063 µg/L, making it one of the most potent animalic materials available. It should be dosed carefully, typically at trace to 1% levels in compounds.
Tenacity
Exceptional. Substantivity on blotter reaches approximately 400 hours at 10% concentration, placing it firmly among the most tenacious perfumery materials. On the Symrise tenacity chart, it persists for months on blotter.
Volatility
Base note. Indoflor sits in the base of a fragrance, providing long-lasting depth and fixative power. Its low vapor pressure (0.006 mmHg @ 25 °C) confirms its classification as a base-note material with excellent staying power.
Applications in Fine Fragrance
Indoflor is a key component for creating white flower notes in modern perfumery. Its indolic character bridges the gap between clean synthetic florals and the complex, sometimes animalic facets found in real flowers like jasmine, tuberose, and orange blossom.
White Floral Accords (traces–0.5%) — Adds depth, naturalness, and an indolic "living flower" quality to jasmine, tuberose, gardenia, and magnolia reconstructions.
Oriental & Amber Compositions (0.1–1.0%) — Contributes warmth, sensuality, and animalic richness. Enhances amber, vanilla, and resinous bases.
Chypre Accords (0.05–0.3%) — Balances overly clean structures with a dose of primal elegance; pairs well with oakmoss, patchouli, and labdanum.
Musky & Animalic Bases (0.1–1.0%) — Reinforces civet, castoreum, and musk reconstructions. Provides a dirty, textured animalic layer.
Leather Accords (traces–0.5%) — Adds the indolic-animalic facet found in classic leather compositions alongside birch tar and isobutyl quinoline.
Performance in Formula
Fixative Power — Outstanding. With a substantivity of ~400 hours, Indoflor acts as a powerful fixative, anchoring lighter top and heart notes.
Radiance Effect — At very low doses, it creates a blooming, radiant effect in floral compositions, adding the characteristic "aliveness" associated with natural flowers.
Stability — Good to excellent in most media. Stability ratings (Symrise): body lotion (good), shampoo (good), soap (good), AP roll-on (very good), powder (very good), APC cleaner (excellent). Poor stability in bleach and citric acid cleaners.
Compatibility — Excellent compatibility with other animalic materials (musks, civet bases, castoreum), white florals (hedione, indole, methyl anthranilate), and woody/balsamic notes.
Solubility — Freely soluble in alcohol and most fragrance solvents. Practically insoluble in water. Non-discoloring in most media.
Industrial & Technical Uses
Functional Perfumery — Widely used in perfumes for soaps, detergents (heavy duty liquids and powders), fabric softeners, and household cleaners where its stability and non-discoloring properties are valued.
Personal Care — Employed in body lotions, shampoos, shower gels, hair conditioners, deodorant sticks, and AP roll-ons. Good stability across a range of pH levels (2.5–10).
Flavor Industry — Not FEMA-listed; no food-grade applications.
Cosmetics — Used as a fragrance ingredient in cosmetic formulations in accordance with EU Regulation 1223/2009 and IFRA guidelines.
Regulatory & Safety Overview
IFRA Status
Restricted. According to the IFRA Standards:
Category 1 (Toys / Lip products): Not suitable
Category 2 (Deodorants / Antiperspirants / Body Spray): Max 1.00%
Category 3 (Eye products): Max 0.50%
Category 4 (Fine fragrance): Not restricted (no upper limit specified)
Recommended use level by Symrise: traces up to 1% in fragrance compounds. Typical industry usage ranges from 0.03% (minimum) to 3.0% (maximum), with an average of 0.3%.
EU Cosmetics Regulation
Permitted for use as a fragrance ingredient in cosmetic products under EU Regulation 1223/2009. Not listed among the 26 EU-regulated fragrance allergens requiring mandatory labeling.
EU REACH
Registered. REACH Registration Number: 01-2120760170-66 (Symrise).
FEMA Status
Not FEMA-listed. Not approved for food/flavor use.
GHS Classification
Signal Word: Warning
Hazard Statements: H361 — Suspected of damaging fertility or the unborn child
Precautionary Statements: P201, P202, P281, P308+P313, P405, P501
Note: PubChem reports that 97.7% of GHS notifications for this substance do not meet hazard criteria. The H361 classification appears in a minority of supplier SDS documents.
Toxicology
Sensitization — RIFM safety assessment (Food Chem Toxicol, 2019; updated 2025) confirms no significant skin sensitization concerns at current declared levels of use.
Phototoxicity — Not expected to be phototoxic or photoallergenic based on UV spectra evaluation.
General — Non-flammable. Flash point >100 °C (TCC). Shelf life: 36 months or longer if stored properly in cool, dry conditions in tightly sealed containers, protected from heat and light.
Environmental Considerations
Biodegradability — Data on the closely related 2,4-dimethyl analog (Magnolan, CAS 27606-09-3) indicates ready biodegradability, suggesting a similar environmental fate for Indoflor.
Bioaccumulation — Low bioaccumulation potential based on the relatively low log P value (1.33–1.84).
Sustainability Credentials
Fully synthetic. Not derived from animal or plant sources. No occurrence in nature.
Related Ingredients on Scentspiracy
Animalic / Indolic Family:
Complementary Materials:
Hedione — Creates a transparent jasmine-floral lift that counterbalances the heaviness of Indoflor
Methyl Anthranilate — Adds a grape-like, orange blossom facet that bridges floral and animalic zones
Magnolan (CAS 27606-09-3) — A structurally related indane-dioxane with a lighter magnolia-floral profile
Cashmeran — Provides a warm, musky-woody complement that extends the base
Related Reading:
References
PubChem, CID 87453 — 4,4a,5,9b-Tetrahydroindeno(1,2-d)-1,3-dioxin
Symrise Perfumery Ingredients — Indoflor® Cryst. Technical Data Sheet (Article No. 660173)
The Good Scents Company — Indoletal (18096-62-3)
RIFM Fragrance Ingredient Safety Assessment, CAS 18096-62-3 (Food Chem Toxicol, 2019; updated 2025)
ChemicalBook — Indolarome Chemical Properties
ECHA eChemPortal — REACH Registration Data
Arctander, S. — Perfume and Flavor Chemicals (Aroma Chemicals), Montclair, NJ