Phenoxanol® / Phenyl Hexanol (55066-48-3) – Technical Ingredient Overview
🏭 Manufacturer — Firmenich (Phenylhexanol), IFF (Phenoxanol®), Givaudan (Mefrosol®)
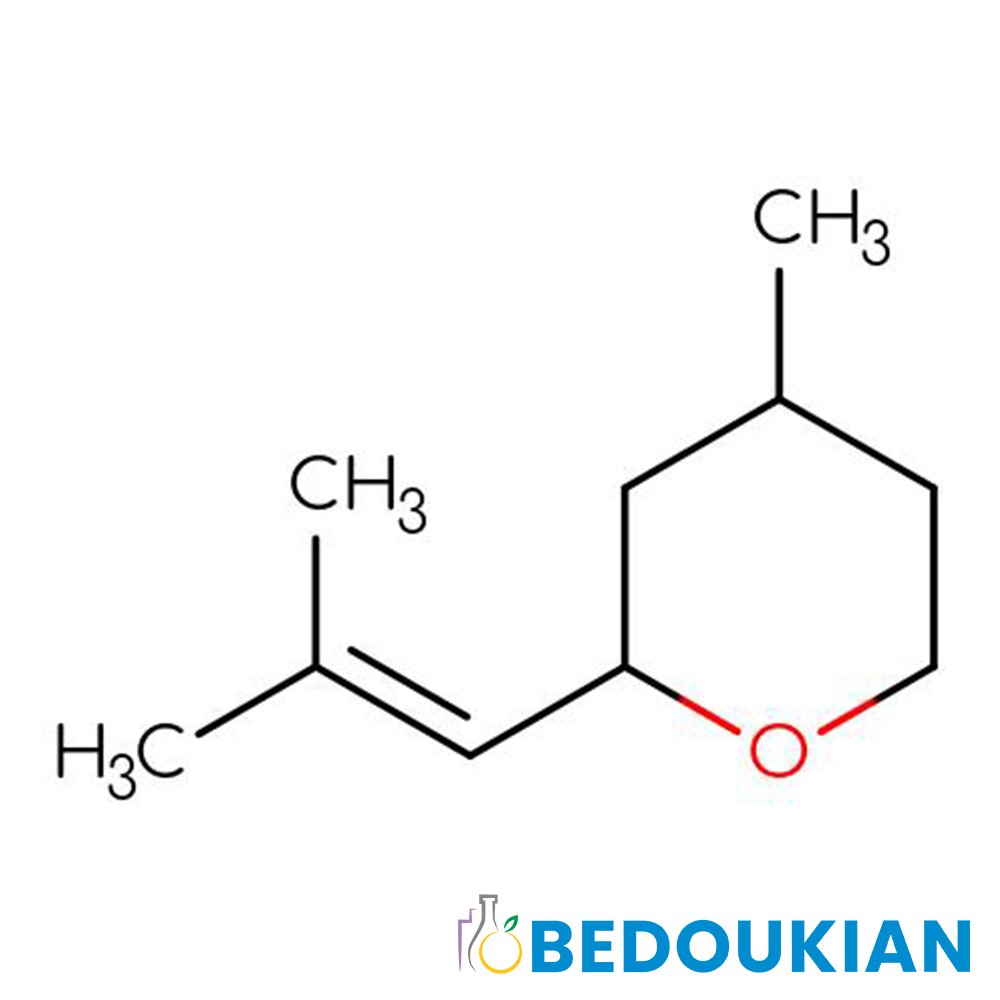
🔎 Chemical Name — 3-Methyl-5-phenylpentan-1-ol
🧪 Synonyms — Phenoxanol®, Phenyl Hexanol, Phenylhexanol, Mefrosol®, Phenoxal, Phenyxol, Phenylisohexanol, γ-Methyl benzene pentanol, Rose absolute pentanol
📂 CAS Number — 55066-48-3
📘 FEMA Number — Not assigned (not used in flavor applications)
⚖️ Molecular Weight — 178.27-178.28 g/mol
🧬 Chemical Formula — C₁₂H₁₈O
📝 Odor Type — Floral (White Floral / Rose-Muguet)
📈 Odor Strength — Medium intensity with exceptional diffusivity and tenacity
👃🏼 Odor Profile — Transparent, soft, fresh white-floral with distinct rose and lily-of-the-valley (muguet) character; light green undertone; aldehydic-aqueous nuances; subtle zesty facet
⚗️ Uses — Long-lasting floral enhancer, muguet compositions, rose alcohol substitute, diffusion extender, Lilial® replacement, white floral modifier
🧴 Appearance — Colorless to pale yellow liquid
What is Phenoxanol®?
Phenoxanol® (Phenyl Hexanol) is a synthetic branched aliphatic alcohol with a phenyl substitution, developed as a high-performance floral component for modern perfumery. Structurally, this molecule serves as an olfactory bridge between phenylethyl alcohol and longer-chain lily alcohols, offering significantly improved diffusivity, substantivity, and tenacity compared to traditional rose alcohols.
The compound is produced through a sophisticated multi-step synthesis: benzaldehyde undergoes Prins cyclocondensation with 3-methyl-3-buten-1-ol in the presence of p-toluenesulfonic acid to yield tetrahydro-4-methylene-5-phenylpyran. This intermediate is then subjected to catalytic hydrogenation, resulting in hydrogenolysis of the benzylic ether bond within the pyran ring to produce 3-methyl-5-phenylpentan-1-ol (Pybus & Sell, 1999; Surburg & Panten, 2006).
The molecule possesses one asymmetric carbon center, technically existing as two enantiomers (3S and 3R configurations). However, both enantiomers exhibit similar olfactory properties, and the racemic mixture is used commercially in perfumery, providing consistent performance across batches.
Unlike more volatile rose alcohols, Phenoxanol's scent is not overpowering but remains evident throughout the entire evaporation curve—from initial spray through complete dry-down. This characteristic makes it a subtle yet highly effective contributor to the structural body and lasting power of fragrances. Its low volatility, high stability, and compatibility with various fragrance bases have made it indispensable in fine fragrance, functional perfumery, and personal care applications.
Historical Background
Phenoxanol® emerged during the mid-20th century as part of the fragrance industry's ongoing search for synthetic alternatives to expensive natural floral materials and molecules with improved performance characteristics. The development was driven by the need for floral alcohols that could provide lasting rose and muguet character without the volatility limitations of phenylethyl alcohol or the cost constraints of natural rose materials.
The synthesis via Prins reaction followed by hydrogenation represented a significant technical achievement, demonstrating how creative organic chemistry could produce molecules with desirable olfactory profiles while maintaining cost-effectiveness and supply reliability. The material was commercialized under multiple tradenames by major fragrance houses: Phenoxanol® by IFF, Phenylhexanol by Firmenich, and Mefrosol® by Givaudan (then Givaudan-Roure), reflecting the competitive development of this valuable molecule.
The compound gained particular prominence as regulatory changes and reformulation requirements in the 2000s-2010s drove perfumers to seek alternatives to restricted materials. Phenoxanol® became especially valuable as a replacement for Lilial® (butylphenyl methylpropional) and Lyral® (hydroxyisohexyl 3-cyclohexene carboxaldehyde) following IFRA restrictions and eventual prohibitions on these popular muguet materials.
The material's transparency and compatibility with modern perfumery techniques—including its stability in both alcohol-based fine fragrances and challenging functional applications—ensured its continued relevance. Its ability to enhance floral compositions without dominating them, combined with exceptional longevity, has made Phenoxanol® a staple ingredient in contemporary perfumery, particularly for white florals, modern green florals, and fabric care applications where lasting softness is desired.
Olfactory Profile
Scent Family: White Floral / Rose-Muguet
Main Descriptors:
Top notes: Fresh, clean floral opening with transparent character; light aldehydic-aqueous quality; subtle green freshness
Heart notes: Soft rose and lily-of-the-valley (muguet) tones develop; waxy-floral nuances emerge; gentle, non-aggressive floral presence
Base notes: Long-lasting diffusive floral character; persistent soft rose; subtle zesty facet distinguishes from other rose/muguet materials (vs. Nerolidol or Rosacetol®)
Intensity: Medium odor strength; soft and non-overpowering, yet noticeably persistent
Tenacity: Exceptional longevity for a floral alcohol; significantly more tenacious than phenylethyl alcohol; remains evident throughout entire evaporation curve; contributes lasting floral lift for hours
Volatility:
Boiling point: 86-91°C at 0.013 kPa
Low volatility contributes to extended performance
Functions across top-heart-base, with primary impact in heart and base notes
Diffusivity: High diffusivity creates lasting floral aura; excellent bloom in both alcohol and functional bases
Character: Pleasant, fresh white-floral odor with exceptional naturalness; transparency ensures compatibility across fragrance families without clashing; green nuance provides freshness; more aldehydic and aqueous compared to structural isomer Majantol®
<aside> <img src="/icons/pencil_yellow.svg" alt="/icons/pencil_yellow.svg" width="40px" />
Related Scentspiracy Materials
Explore complementary white-floral and rosy materials:
Citronellol - Rosy-floral monoterpenoid alcohol
Rosalva - Fresh, aldehydic-dewy rose character
Phenirat - Fruity-floral ester with rose-honey notes </aside>
Applications in Fine Fragrance
Phenoxanol® serves multiple essential functions in modern perfumery:
Primary Applications:
Muguet (Lily-of-the-Valley) compositions: Essential component and Lilial® replacement
Rose accords: Replaces or supplements phenylethyl alcohol and rose alcohols with improved longevity
Peony and white florals: Provides transparent floral character without heaviness
Modern green florals: Contributes fresh, natural floral-green notes
Light fruity-floral blends: Adds subtle floral depth to fruit-forward compositions
Diffusion extension: Enhances lasting power across various fragrance types
Notable Pairings:
Lily alcohols and muguet aldehydes: Hydroxycitronellal, Bourgeonal®, Lilial® (historically), Lysmeral®
Rose materials: Citronellol, geraniol, phenylethyl alcohol, rose oxide
Floral alcohols: Linalool, terpineol, benzyl alcohol
Green materials: Cis-3-hexenol, violet leaf, galbanum notes
Modern synthetics: Works harmoniously in contemporary transparent structures
Typical Usage Levels:
Fine fragrance: Effective at ~1%, tolerates up to 20% in high-floral compositions without causing imbalance
Functional fragrances: 1-5% for lasting floral softness in fabric softeners, laundry, haircare
Personal care: 0.5-3% in lotions, creams, body care for gentle floral character
Performance in Formula
Phenoxanol® exhibits excellent formulation characteristics:
Physical Properties:
Density: 0.960-0.964 g/mL at 20°C
Refractive index: nD²⁰ 1.511-1.514
Boiling point: 86-91°C at 0.013 kPa
Appearance: Colorless to pale yellow liquid
Solubility & Compatibility:
Readily soluble in ethanol and oil-based systems
Low reactivity ensures broad compatibility
Performs well in both leave-on and rinse-off formulations
Excellent blendability with diverse perfumery materials
Stability Profile:
High stability: Maintains performance under standard storage
Oxidative stability: Good resistance to oxidation
pH tolerance: Generally stable, though unstable in acid cleaners and very alkaline products
Color stability note: Benzene ring can cause slight yellowing over time (common with aromatic materials)
Performance Characteristics:
Exceptional tenacity supports long-lasting floral tone
Low volatility provides extended release
Gentle odor profile suitable for controlled sensory compositions
Supports lasting softness in fabric care applications
Industrial & Technical Uses
Beyond fine fragrance, Phenoxanol® finds extensive application in:
Perfumery:
Fine fragrances (white florals, muguet, soft chypres, modern green florals)
Functional perfumery (fabric softeners, laundry detergents, powder detergents)
Haircare (shampoos, conditioners for lasting floral softness)
Personal Care:
Creams, lotions, body care products
Deodorants and antiperspirants
Leave-on formulations benefit from its gentle, persistent character
Household Products:
Fabric care (softeners, detergents) where long-lasting floral softness is desired
Body care formulations
Products requiring stable, non-aggressive floral character
Regulatory & Safety Overview
IFRA Status:
Not restricted under IFRA Amendment 51 (notified June 30, 2023)
Permitted for use in all fragrance product categories without specific concentration limits
EU Cosmetics Regulation 1223/2009:
Not listed among the 26 declarable allergens
EC Number: 259-461-3
Compliant for cosmetic use
ECHA (REACH) Registration:
Registered substance under EU REACH
Not classified as hazardous
Multi-constituent substance; both enantiomers registered
Safety Profile:
Non-sensitizing at standard use concentrations
No evidence of skin irritation in typical applications
Low volatility, high stability
Biodegradable: Not classified as PBT (Persistent, Bioaccumulative, Toxic) or vPvB (very Persistent, very Bioaccumulative)
Compatible with ethanol and oil-based formulations
Generally well-tolerated in both fine fragrance and functional applications
Storage & Handling:
Store in tightly closed containers
Protect from light to minimize yellowing
Cool, dry storage conditions recommended
Stable under normal use and storage conditions
References
Pybus, D. H., & Sell, C. S. (Eds.). (1999). The chemistry of fragrances: From perfumer to consumer. Cambridge: Royal Society of Chemistry.
Surburg, H., & Panten, J. (2006). Common fragrance and flavor materials: Preparation, properties and uses (5th ed.). Weinheim: Wiley-VCH. https://doi.org/10.1002/3527608214