Musk Ketone - Technical Ingredient Overview
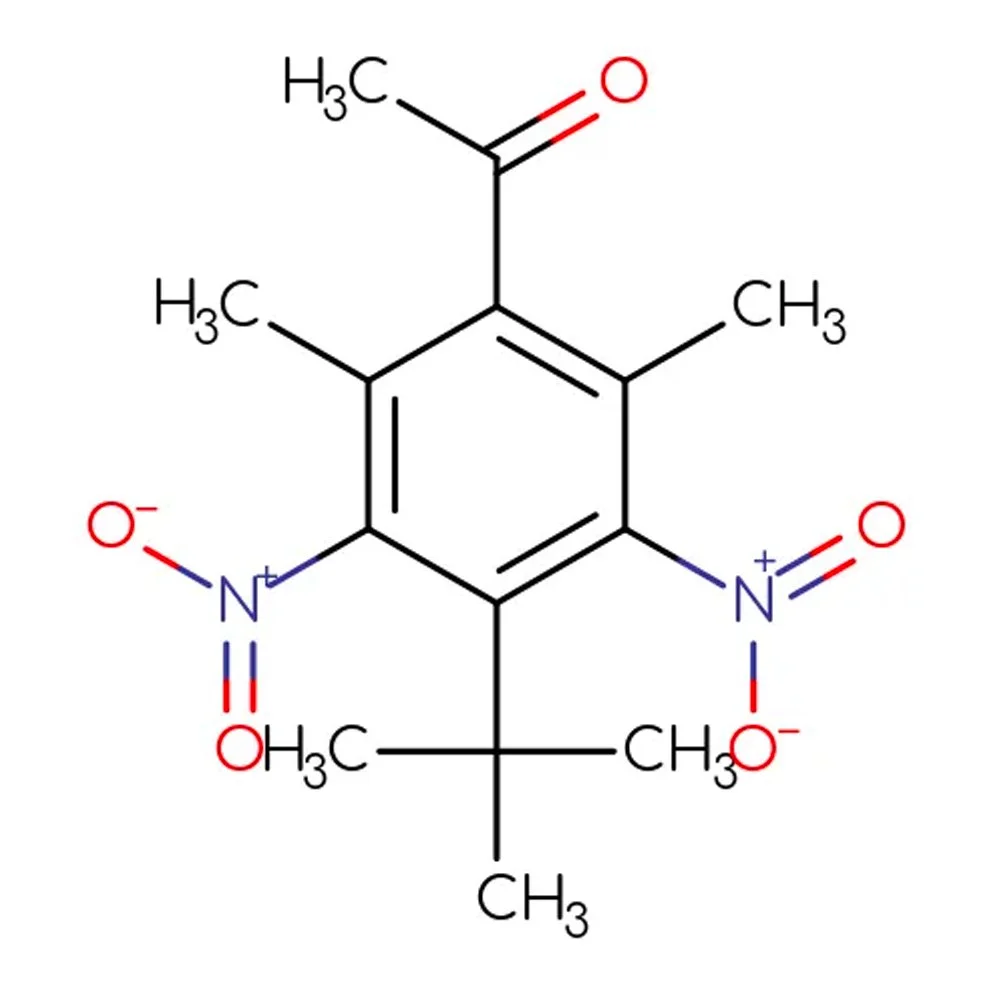
🔎 Chemical Name — 4-tert-Butyl-2,6-dimethyl-3,5-dinitroacetophenone (NIST WebBook, 2025)
🧪 Synonyms — Ketone Moschus; 3,5-Dinitro-2,6-dimethyl-4-tert-butyl acetophenone; 2,6-Dimethyl-3,5-dinitro-4-tert-butylacetophenone; 1-[4-(1,1-Dimethylethyl)-2,6-dimethyl-3,5-dinitrophenyl]ethanone (PubChem, 2025)
📂 CAS Number — 81-14-1 (PubChem, 2025)
📘 FEMA Number — Not assigned (not approved as a flavoring substance)
⚖️ Molecular Weight — 294.30 g/mol (PubChem, 2025)
📝 Odor Type — Nitro musk
📈 Odor Strength — Very strong, highly tenacious
👃🏼 Odor Profile — Sweet, warm, powdery musk with subtle animalic undertones; very persistent with a soft, slightly floral-fruity character; less floral than Musk Ambrette; exhibits exceptional longevity and substantivity (Burdock, 2016; Rowe, 2005)
⚗️ Uses — Fixative in fine fragrance, particularly in floral, oriental, amber, aldehydic, leather, and chypre compositions; historically used in soap and cosmetic perfumes (Burdock, 2016)
🧴 Appearance — Yellowish to pale yellow crystalline solid or platelets; melting point 134-137°C (Sigma-Aldrich, 2025; NIST WebBook, 2025)
What is Musk Ketone?
Musk Ketone is a synthetic nitroaromatic compound belonging to the nitro musk family of fragrance ingredients. Chemically classified as a dinitrophenyl ketone, it represents one of the earliest successful attempts to replicate the olfactory characteristics of natural animal musk through synthetic chemistry (Baur, 1898; Rowe, 2005).
Unlike natural musks derived from animal sources or macrocyclic musks that mimic natural musk structures, Musk Ketone is an aromatic compound featuring two nitro groups (-NO₂) attached to a benzene ring, along with methyl and tert-butyl substituents. This unique structure gives it a distinctive sweet, powdery musk character with remarkable tenacity and fixative properties (Kraft, 2000).
The compound does not occur in nature and is entirely synthetic in origin. It forms part of the first generation of synthetic musks that revolutionized perfumery in the late 19th and early 20th centuries (Burdock, 2016).
Historical Background
The discovery of Musk Ketone traces back to pioneering work by German chemist Albert Baur in the late 19th century. In 1888, while attempting to develop more effective explosives based on trinitrotoluene (TNT), Baur serendipitously discovered that certain nitrated aromatic compounds possessed powerful musk-like odors (Baur, 1889; American Chemical Society, 2022).
Baur's first synthetic musk, known as "Musk Baur" or "Tonquinol," was patented in 1889 and created immediate commercial interest as an affordable alternative to natural Tonkin musk, which at the time cost approximately $1,000 per kilogram (David, 2017). Following this breakthrough, Baur continued his research into nitrated aromatic compounds.
In 1894, Baur patented Musk Ketone, synthesized through the Friedel-Crafts acetylation of 1,3-dimethyl-5-tert-butylbenzene followed by nitration with nitric acid (Baur, 1898; Bedoukian, 1967). This compound was said to resemble natural deer musk more closely than his earlier discoveries and quickly became one of the most important synthetic musks in perfumery (NCBI Bookshelf, 1998).
Throughout the early to mid-20th century, Musk Ketone featured prominently in landmark fragrances, including Fougère Royale (1882, though Musk Ketone was incorporated in later versions), Chanel No. 5 (1921), and continued into modern classics such as Musk Ravageur (2000) by Frederic Malle and Ambre Sultan (1993) by Serge Lutens, where its warm, powdery character contributed significantly to their base notes (Fragrantica, 2011; Kraft et al., 2007).
By the 1990s, annual worldwide production of nitro musks had declined significantly due to environmental and toxicological concerns. Production shifted primarily to Musk Ketone (approximately 21% of nitro musk production) and Musk Xylene (67%), with other nitro musks being phased out (NCBI Bookshelf, 1998).
Olfactory Profile
Scent Family
Nitro Musk — Part of the aromatic musk classification, specifically within the nitroaromatic sub-category
Main Descriptors
Musk Ketone delivers a distinctively sweet, warm, and powdery musk character with notable depth and complexity. The odor profile includes:
Primary character: Soft, clean musk with pronounced powdery qualities
Secondary facets: Subtle animalic undertones, discrete floral-fruity nuances. Slightly ambery, warm aromatic aspects
Overall impression: Smooth, sensual, tenacious, with exceptional substantivity
The scent is generally considered less floral and more linear in its evolution compared to Musk Ambrette, offering a cleaner and more powdery drydown (PerfumersWorld, 2025; Rowe, 2005).
Intensity
Very Strong — Musk Ketone possesses exceptional odor strength and is effective even in trace amounts, making it highly valued as a fixative material.
Tenacity
Exceptional — One of the longest-lasting synthetic musks available, with persistence measured in days rather than hours. Its low volatility and high substantivity on both skin and textiles contribute to its remarkable longevity (Bois de Jasmin, 2005).
Volatility
Base Note — Classified as a heavy base note material with extremely low volatility. The compound exhibits minimal evaporation at room temperature, ensuring sustained presence throughout the fragrance's lifecycle.
Fixative Role
Musk Ketone functions as a powerful fixative, stabilizing volatile top and middle notes while imparting warmth, depth, and roundness to fragrance compositions. It enhances the overall tenacity of perfume formulas and helps harmonize disparate elements within complex accords (American Chemical Society, 2022).
Applications in Fine Fragrance
Musk Ketone historically played a central role in structuring floral bouquets, oriental compositions, chypres, aldehydic florals, leather fragrances, and amber accords. Its primary function was to provide a warm, powdery musk foundation that enhanced longevity and added sensuality to fragrances.
Typical pairings and accords:
Aldehydic florals: Combines with aldehydes and floral absolutes to create powdery, sophisticated compositions
Oriental bases: Blends with labdanum, vanilla, benzoin, and amber notes to reinforce warmth and depth
Chypre structures: Provides musky drydown beneath oakmoss, patchouli, and bergamot
Floral compositions: Enhances gardenia, mimosa, jasmine, and honeysuckle accords
Animalic notes: Pairs with costus root, curcuma, civet replacers, and castoreum for richer, more complex bases
Due to regulatory restrictions and environmental concerns, Musk Ketone has been largely replaced by macrocyclic musks (e.g., Exaltolide, Ethylene Brassylate) and polycyclic musks (e.g., Galaxolide, Tonalide) in modern formulations (Rowe, 2005).
Performance in Formula
Musk Ketone exhibits excellent solubility in alcohol and most organic solvents commonly used in perfumery, including ethanol, dipropylene glycol (DPG), and benzyl benzoate. However, it can be challenging to dissolve at room temperature due to its crystalline nature, often requiring gentle heating or extended stirring (Fraterworks, 2024).
In compositions, it demonstrates remarkable stability once dissolved and blends seamlessly with both natural and synthetic materials. Its high molecular weight and low vapor pressure contribute to its fixative properties, significantly extending the longevity of more volatile components.
Key performance characteristics:
Blending behavior: Harmonizes well with florals, woods, spices, and resins
Dosage impact: Effective at concentrations as low as 0.1-1%; higher concentrations (up to 5-10% in some vintage formulas) intensify its presence
Stability: Highly stable in finished products, though photodegradation can occur with prolonged light exposure
Industrial & Technical Uses
Beyond fine fragrance, Musk Ketone found extensive use in functional perfumery including:
Soap perfumes and detergent formulations
Cosmetic products (creams, lotions, shampoos)
After-shave lotions
Household care products
Its tenacity on fabrics made it particularly valuable for laundry products, where long-lasting fragrance was desired. However, these applications have significantly declined since the 1990s due to regulatory restrictions and the availability of safer alternatives (NCBI Bookshelf, 1998; Lew, 2023).
Regulatory & Safety Overview
IFRA Status
Specification Standard (IFRA Standard published 2010, confirmed in 51st Amendment, 2023)
Musk Ketone is permitted for use under IFRA regulations but subject to specific requirements:
Must contain less than 0.1% Musk Xylene as an impurity (Musk Xylene itself is prohibited)
Some jurisdictions limit the total concentration in finished perfumes to 1.4%
IFRA Certificate: IFRA Standards - 51st Amendment
EU Cosmetics Regulation (EC 1223/2009)
Restricted — Listed in Annex III, Entry 97 of EU Cosmetics Regulation
Musk Ketone is classified as a CMR Category 2 substance (Carcinogenic, Mutagenic, or toxic for Reproduction - Category 2) under EU regulations. It is permitted for use in cosmetic products only under specific conditions and concentration limits as evaluated and approved by the Scientific Committee on Consumer Safety (SCCS) (European Commission, 2015).
FEMA Status
Not Assigned — Musk Ketone is not approved for use as a flavoring substance and does not have FEMA (Flavor and Extract Manufacturers Association) GRAS (Generally Recognized as Safe) status.
Toxicology
Studies have provided evidence of lack of genotoxic potential for Musk Ketone. However, it has been identified as a strong inducer of phase I enzymes in rodents and a co-genotoxicant in vitro in human-derived cells at relatively low doses. This suggests that exposure to Musk Ketone might increase susceptibility to health hazards caused by other carcinogens (ChemicalBook, 2025; Schmeiser et al., 2001).
Due to its low biodegradability, Musk Ketone has been detected in aquatic environments (surface water, sediments, fish) and has been shown to concentrate in human fatty tissue and breast milk, indicating constant human exposure. These bioaccumulation concerns have driven regulatory scrutiny and industry movement toward alternative musk ingredients (NCBI Bookshelf, 1998).
References
American Chemical Society. (2022). Musk ketone - Molecule of the Week. Retrieved from https://www.acs.org/molecule-of-the-week/archive/m/musk-ketone.html
Baur, A. (1889). German Patent (DRP) for synthetic musk compounds.
Baur, A. (1898). Ueber Keton-Moschus. Berichte der Deutschen Chemischen Gesellschaft, 31, 1344-1349.
Bedoukian, P. Z. (1967). Perfumery synthetics and isolates. Van Nostrand.
Bois de Jasmin. (2005). Musk in fragrance: Salt and butter of perfumery. Retrieved from https://boisdejasmin.typepad.com
Burdock, G. A. (2016). Fenaroli's handbook of flavor ingredients (6th ed.). CRC Press.
ChemicalBook. (2025). Musk ketone (81-14-1). Retrieved from https://www.chemicalbook.com
David, B. (2017). Artificial nitromusks, stories of chemists and businessmen. European Journal of Organic Chemistry, 2017(6), 1108-1134. https://doi.org/10.1002/ejoc.201601249
European Commission. (2015). Commission guidance document on the application of Article 31(1)(f) of Regulation (EC) No 1223/2009. Retrieved from https://ec.europa.eu
Fragrantica. (2011). Natural and synthetic musk. Retrieved from https://www.fragrantica.com/news/Natural-and-Synthetic-Musk-5243.html
Fraterworks. (2024). Musk Ketone product information. Retrieved from https://fraterworks.com
IFRA (International Fragrance Association). (2023). IFRA standards – 51st amendment. Retrieved from https://ifrafragrance.org
Kraft, P. (2000). Aroma chemicals IV: Musks. In D. J. Rowe (Ed.), Chemistry and technology of flavors and fragrances(pp. 143-168). Blackwell Publishing.
Kraft, P., Ledard, C., & Goutell, P. (2007). From Rallet N°1 to Chanel N°5 vs. Mademoiselle Chanel N°1. Perfumer & Flavorist, 32(October), 36-48.
Lew, D. (2023). Polycyclic musks - Nitro musk compounds. Retrieved from https://www.drdarrinlew.us
NCBI Bookshelf. (1998). Musk ambrette and musk xylene. In Printing processes and printing inks, carbon black and some nitro compounds. IARC Monographs. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK424348
NIST WebBook. (2025). Musk ketone.
PerfumersWorld. (2025). Musk Ketone technical data. Retrieved from https://www.perfumersworld.com
PubChem. (2025). Musk ketone (Compound CID 6669). National Center for Biotechnology Information. Retrieved from https://pubchem.ncbi.nlm.nih.gov
Rowe, D. J. (Ed.). (2005). Chemistry and technology of flavors and fragrances. Blackwell Publishing.
Schmeiser, H. H., Gminski, R., & Mersch-Sundermann, V. (2001). Evaluation of health risks caused by musk ketone. International Journal of Hygiene and Environmental Health, 203(4), 293-299.
Sigma-Aldrich. (2025). Musk ketone product specifications. Retrieved from https://www.sigmaaldrich.com