🌸 Jasmolactone (CAS 32764-98-0, FEMA 4441, EC 251-201-7) is a synthetic δ-lactone with a rich, natural jasmine-petal character and a creamy peach-apricot-coconut body. Commercialised chiefly by dsm-firmenich as Jasmolactone Delta (code 965414), it is a staple for white-floral reconstructions, lactonic-fruity florals, tea accords and solar-tropical compositions, typically dosed at 0.1–1% of the concentrate.
Jasmolactone (CAS 32764-98-0) — technical ingredient overview
🏭 Manufacturer — dsm-firmenich (JASMOLACTONE DELTA, code 965414). Also produced or distributed by Bedoukian Research (#413 Jasmolactone Extra C), Vigon International, Sigma-Aldrich / Merck (fragrance grade), Biosynth, Chem-Impex.
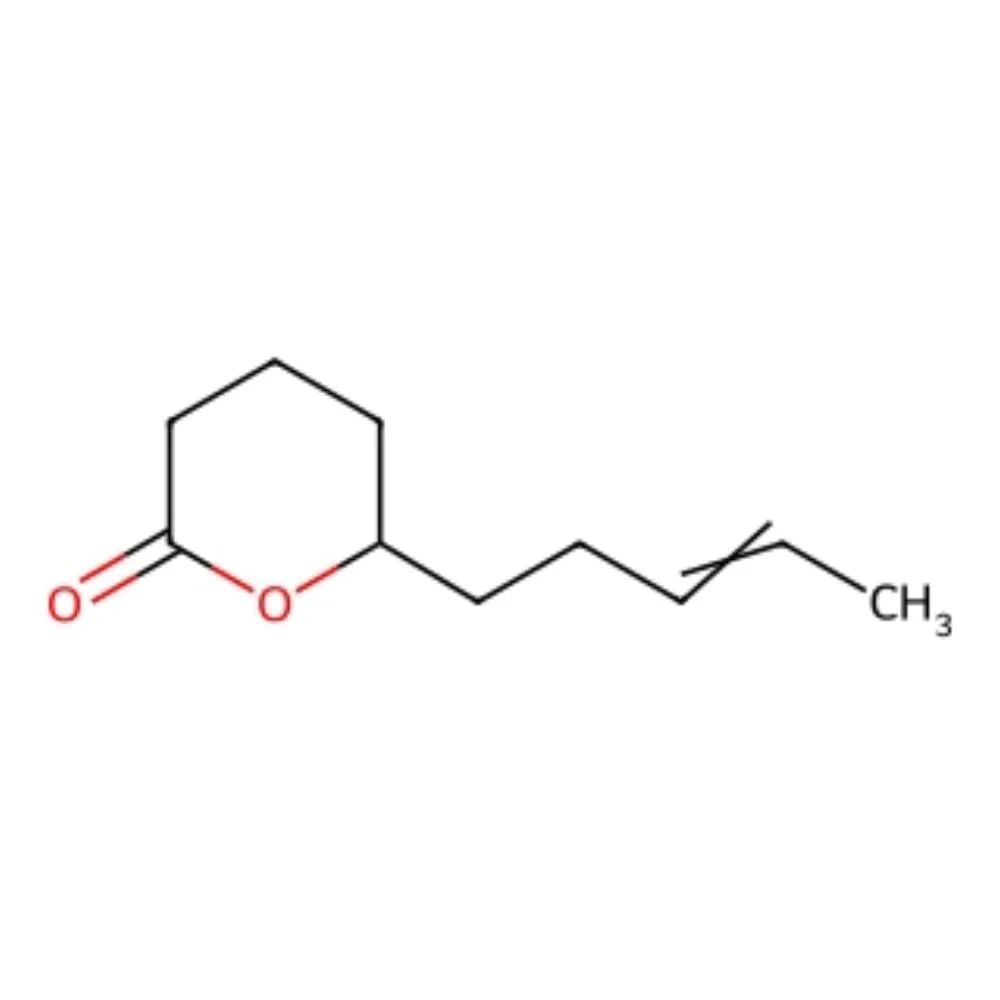
🔎 Chemical name (CAS) — 2H-Pyran-2-one, tetrahydro-6-(3-pentenyl)-
🧬 IUPAC name — 6-(pent-3-en-1-yl)tetrahydro-2H-pyran-2-one
🧪 Synonyms — δ-Jasmolactone; Jasmolactone Delta; Jasmolactone Extra; Petal Pyranone; 5-hydroxydec-8-enoic acid δ-lactone.
📂 CAS Number — 32764-98-0
📘 FEMA Number — 4441 (GRAS)
🔢 EC Number — 251-201-7
🗂️ FLAVIS Number — 10.040
🗂️ JECFA Number — 1994
🗂️ REACH Number — 01-2120042409-58
⚖️ Molecular Weight — 168.23 g/mol
🔬 Molecular Formula — C₁₀H₁₆O₂
📝 Odor Type — Floral-lactonic
📈 Odor Strength — High (active at 0.1–1%)
👃🏼 Odor Profile — Rich, natural jasmine petal with delicate fruitiness of peach, apricot and coconut milk; creamy, slightly waxy-fatty with a soft tropical floral radiance. A faint celery/hay nuance may appear in dilution.
⚗️ Uses — Fine fragrance (white florals, tea, ambery-floral, solar), functional perfumery, flavors.
🧴 Appearance — Colourless to pale-yellow liquid.
Additional physical properties
🌡️ Melting Point — Not reported in primary literature (liquid at room temperature).
🔥 Boiling Point — 283–301 °C at 760 mmHg (Vigon, Chem-Impex, The Good Scents Co.); ~150 °C at reduced pressure.
💧 Density (20 °C) — ~1.01 g/cm³
🔍 Refractive Index (n20/D) — 1.475 – 1.480
💧 Solubility — Fully soluble in ethanol, DPG, IPM and common perfumery solvents; practically insoluble in water.
🔄 Specific Rotation — Commercial material is racemic; no optical rotation typically specified.
✅ Purity — ≥95% (fragrance grade; Bedoukian ≥96% sum of isomers).
💨 Vapor Pressure — 0.0022 mmHg at 20 °C
🔥 Flash Point — 110 °C (230 °F), closed cup.
📦 Typical Shelf Life — 36 months in sealed, light- and heat-protected container (Vigon TDS).
🌱 Biogenic Carbon Content — Not declared; synthetic origin, produced under green-chemistry principles (dsm-firmenich Eco Ingredient Compass®).
What is Jasmolactone?
Jasmolactone is a synthetic δ-lactone — a six-membered 2H-pyran-2-one ring — bearing an unsaturated pentenyl side chain. It belongs to the broader "jasmine-lactone" family that reproduces the creamy, petal-like facet found in the headspace of Jasminum grandiflorum and Jasminum sambac. In nature the molecule occurs only in trace amounts (notably reported in Osmanthus absolute), quantities far too small for economic isolation: all commercial supply is produced by chemical synthesis, typically via intramolecular esterification of 5-hydroxydec-8-enoic acid (or an equivalent hydroxy-unsaturated acid) catalysed by acid, followed by purification by fractional distillation.
It is a positional isomer of Jasminlactone / Jasmine Lactone (CAS 25524-95-2), from which it must be carefully distinguished: the two molecules differ only in the position of the double bond on the pentenyl chain, yet they have noticeably different olfactory signatures and separate regulatory dossiers.
Historical background
Jasmolactone was introduced to perfumery around 1961, during the wave of lactonic materials that followed the industrial commercialisation of γ- and δ-decalactones. Firmenich (today dsm-firmenich) developed and marketed it under the trade name Jasmolactone Delta (product 965414), and it rapidly became a staple in the reconstruction of jasmine and other white florals. Over the following decades, complementary producers and resellers — Bedoukian, Vigon, Sigma-Aldrich, Biosynth, Chem-Impex — brought equivalent grades to the market. The molecule is closely related to Jasmine Lactone / Jasminlactone (CAS 25524-95-2) but remains chemically and olfactively distinct.
Olfactory profile
Scent family
Floral-lactonic, with pronounced jasmine petal character and fruity-creamy facets (peach, apricot, coconut milk). Secondary descriptors include waxy, fatty, milky and subtly tropical.
Main descriptors
Jasmine petal · peach · apricot · coconut milk · creamy · fruity · floral · waxy-lactonic · soft tropical. A discreet celery/hay nuance appears in high dilution (a marker dsm-firmenich explicitly lists among its technical descriptors).
Intensity
High. Even at 0.1–1% of the concentrate it reshapes white-floral accords, giving natural "petal" radiance and a creamy body. It behaves in a balanced, predictable way and is easy to place in a composition. Overdosed, it becomes fatty-waxy and can flatten jasmine absolutes.
Tenacity
Excellent. On a smelling strip the material lasts well over 48 hours; several suppliers report it can still be detected after up to two weeks, making it an effective natural-style fixative for white florals.
Volatility
Heart-to-base note. ScenTree classifies it as a base (volatility), while most formulation references treat it as a long-lasting heart note modifier with fixative behaviour.
Applications in fine fragrance
Jasmolactone is most commonly used between 0.1% and 1% of the concentrate, with a typical working benchmark around 0.3–0.5% — high enough to reshape an accord, low enough to avoid waxy drift.
White-floral reconstructions (0.3–1%) — jasmine, tuberose, gardenia, frangipani, ylang and osmanthus: it "naturalises" synthetic bouquets and adds the petal/creamy facet typical of fresh blossoms.
Lactonic-fruity floral accords (0.1–0.5%) — coupled with γ-undecalactone, δ-decalactone, Prunolide and peach/apricot materials, it reinforces the fruit-through-flower effect of modern florals.
Tea and ambery-floral accords (0.05–0.3%) — small doses bring a petal facet to tea notes and a creamy roundness to ambery-floral structures.
Tropical / solar accords (0.1–0.7%) — coconut-milk and monoï-style effects, especially in synergy with Methyl Laitone, Coconut Aldehyde, salicylates and ylang.
Gourmand & milky accords (0.05–0.3%) — softens gourmand blends with a natural creaminess that avoids the "candy" effect of pure vanillics.
How perfumers use Jasmolactone
Start low, then scale up. Begin at 0.1% of the concentrate and evaluate at 24 h and 72 h. Jasmolactone's character builds over time.
Pair it with Hedione®. Hedione gives the airy diffusion; Jasmolactone delivers the creamy petal body. Together they reconstruct the "living jasmine" effect that neither achieves alone.
Use it to naturalise synthetic jasmines. A 0.3–0.5% dose on a synthetic jasmine base instantly adds the missing "fresh petal" signal.
Balance with fruity-lactonic partners. γ-Undecalactone, δ-decalactone and Prunolide extend the peach/apricot body; salicylates add warmth.
Watch for overdose. Above ~1.5% the molecule turns waxy-fatty and may dull the top notes; always re-check against a control without Jasmolactone.
Great for stable bases. Because of its low volatility and stability in anhydrous media, it performs well in solid perfumes, oil-based perfumes and alcohol-free body products.
Performance in formula
Fixative power — Medium-high; extends the longevity of white-floral and fruity-floral accords.
Radiance effect — High; provides a "petal glow" that lifts Hedione-rich bouquets.
Stability — Good in alcoholic solutions; like all lactones, it may slowly polymerise over long storage, increasing viscosity and occasionally causing a phase shift in ethanolic solutions. Store cool, dark and in tightly sealed containers.
Compatibility — Blends seamlessly with Hedione®, rose ketones (α/β damascones, damascenone), ethyl safranate, indole, methyl jasmonate, cis-jasmone, other lactones, salicylates, sandalwood materials and coconut aldehyde.
Solubility — Fully soluble in DPG, ethanol and common carriers at working concentrations.
Industrial & technical uses
Functional perfumery — Fabric softeners, detergents, shampoos and body care where a creamy-floral lift is desired.
Personal care — Fine body lotions, solid perfumes and anhydrous systems (excellent stability in oil-based matrices).
Flavor industry — FEMA 4441 / JECFA 1994 / FLAVIS 10.040: used as a flavouring agent for peach, apricot, coconut, tropical-fruit and dairy notes, at typical self-limiting levels.
Cosmetics — Permitted under EU Cosmetics Regulation 1223/2009 as a fragrance ingredient.
Regulatory & safety overview
IFRA status
No restriction under the current IFRA 51st Amendment (Firmenich IFRA Certificate of Conformity for product 965414, issued 16 October 2024; revised 10 November 2023). Jasmolactone is neither on the IFRA Prohibited nor on the IFRA Restricted list for any product category.
EU Cosmetics Regulation
Permitted ingredient under Regulation (EC) No. 1223/2009. Not listed among the EU-regulated fragrance allergens that require on-pack labelling.
EU REACH
Fully registered under REACH registration number 01-2120042409-58 (dsm-firmenich dossier).
FEMA status
GRAS (Generally Recognised As Safe) under FEMA 4441 for flavour use at typical self-limiting levels. Also JECFA-evaluated under number 1994.
GHS classification
Generally not classified as a dangerous substance for transport. Typical supplier SDSs address skin/eye irritation precautions and the standard "avoid direct contact" handling rules; always refer to the supplier's current SDS.
Toxicology (RIFM 2022 safety assessment)
According to the RIFM fragrance ingredient safety assessment for tetrahydro-6-(3-pentenyl)-2H-pyran-2-one (Api et al., Food Chem. Toxicol., 2022; PMID 34864142):
Genotoxicity — Not genotoxic.
Repeated-dose, reproductive and local respiratory toxicity — No NOAEL/NOAEC available; exposure is below the TTC (Threshold of Toxicological Concern).
Skin sensitisation — Not a concern for skin sensitisation at current declared use levels.
Phototoxicity / photoallergenicity — Not expected to be phototoxic or photoallergenic (UV/Vis spectra show no absorption above 290 nm).
Conclusion — Safe as used under current conditions.
Environmental considerations
Persistence — Readily biodegradable: measured 87% in OECD 301F (RIFM, 2012). dsm-firmenich's Eco Ingredient Compass® reports 50–70% biodegradation range in standardised tests.
Bioaccumulation — Low potential. Screening-level BCF 16.56 L/kg (EPI Suite v4.11).
Ecotoxicity — Screening-level fish LC50 = 124.4 mg/L (RIFM Framework).
PBT/vPvB — Not PBT or vPvB per IFRA Environmental Standards.
Sustainability credentials
dsm-firmenich rates JASMOLACTONE DELTA in its Eco Ingredient Compass® as readily biodegradable with a favourable green score; synthesis follows green-chemistry principles where possible. No renewable-carbon certification (e.g. ASTM D6866 / TÜV biobased) is currently published by the manufacturer.
References
Api, A.M. et al., RIFM fragrance ingredient safety assessment, tetrahydro-6-(3-pentenyl)-2H-pyran-2-one, CAS Registry Number 32764-98-0, Food Chem. Toxicol., 2022, 159 Suppl. 1, 112734. PMID 34864142. https://pubmed.ncbi.nlm.nih.gov/34864142/
Fragrance Material Safety Resource (Elsevier), 2H-pyran-2-one, CAS 32764-98-0 — RIFM safety assessment PDF. https://fragrancematerialsafetyresource.elsevier.com/sites/default/files/32764-98-0.pdf
Api, A.M. et al., RIFM fragrance ingredient safety assessment, 5-hydroxy-7-decenoic acid δ-lactone (Jasminlactone), CAS 25524-95-2, Food Chem. Toxicol., 2022. https://www.sciencedirect.com/science/article/abs/pii/S0278691522000503
PubChem, Jasmine lactone, CID 5352626. National Library of Medicine / NIH. https://pubchem.ncbi.nlm.nih.gov/compound/Jasmine-lactone
PubChem, 8-Decen-5-olide / Jasmolactone, CID 6438319. https://pubchem.ncbi.nlm.nih.gov/compound/6438319
dsm-firmenich Studio, Jasmolactone Delta (PE 965414) technical sheet. https://studio.dsm-firmenich.com/product/jasmolactone-delta-pe-965414
dsm-firmenich Studio, Jasminlactone (PE 952700) technical sheet. https://studio.dsm-firmenich.com/product/jasminlactone-pe-952700
Firmenich, IFRA Certificate of Conformity for JASMOLACTONE 965414, up to and including IFRA 51st Amendment, issued 16 October 2024. https://perfumersupplyhouse.com/wp-content/uploads/2015/06/965414_JASMOLACTONE-965414_4102_US_EN.pdf
IFRA, IFRA Standards 51st Amendment — Standards Documentation. https://ifrafragrance.org/standards-library
Sigma-Aldrich / Merck, Jasmolactone ≥95% (W444165). https://www.sigmaaldrich.com/US/en/product/aldrich/w444165
Vigon International, Jasmolactone TDS (Item #502805). https://www.vigon.com/product/jasmolactone/
Bedoukian Research, #413 Jasmolactone Extra C catalogue. https://www.thegoodscentscompany.com/data/rw1000391.html
The Good Scents Company, Petal Pyranone / Jasmolactone Delta (32764-98-0). https://www.thegoodscentscompany.com/data/rw1459461.html
Fragrance Conservatory, Tetrahydro-6-(3-pentenyl)-2H-pyran-2-one. https://fragranceconservatory.com/ingredient/tetrahydro-6-3-pentenyl-2h-pyran-2-one
Perfumer Supply House, Jasmolactone (Firmenich) — SDS, IFRA 51 and allergen documents. https://perfumersupplyhouse.com/product/jasmolactone-firmenich/
Biosynth, Jasmolactone (HBA76498, CAS 32764-98-0). https://www.biosynth.com/p/HBA76498/32764-98-0-jasmolactone
ECHA, REACH registered-substance database (EC 251-201-7). https://echa.europa.eu/
FEMA Flavor & Extract Manufacturers Association, GRAS No. 4441. https://www.femaflavor.org/
EU Regulation (EC) No. 1223/2009 on cosmetic products. https://eur-lex.europa.eu/eli/reg/2009/1223/oj
Arctander, S., Perfume and Flavor Chemicals (Aroma Chemicals), Vol. I–II, 1969 — entries on lactones and jasmine materials.
Surburg, H.; Panten, J., Common Fragrance and Flavor Materials, 6th ed., Wiley-VCH, 2016 — chapter on lactones.