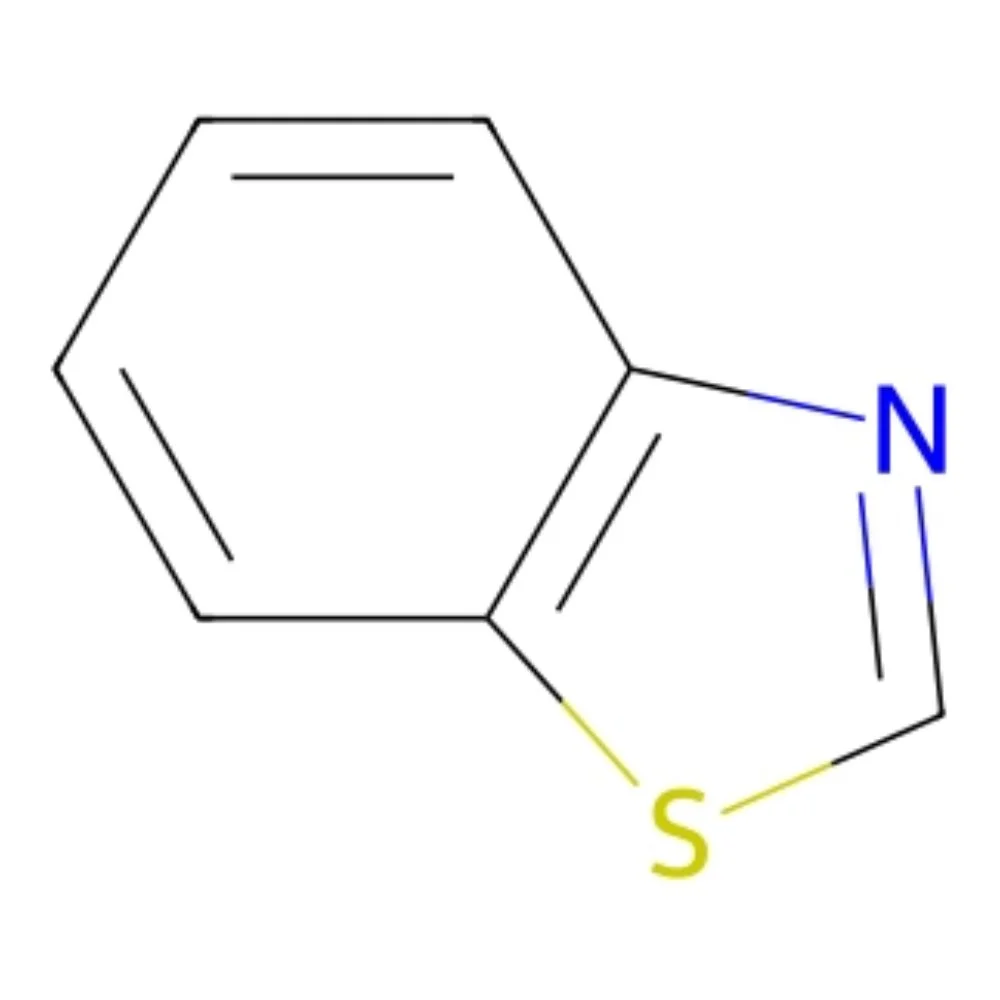
Benzothiazole (CAS 95-16-9) – Technical Ingredient Overview
🔎 Chemical Name — 1,3-Benzothiazole
🧪 Synonyms — Benzosulfonazole; 1-Thia-3-azaindene; Vangard BT; USAF EK-4812; o-2857; Benzothiazol
📂 CAS Number — 95-16-9
📘 FEMA Number — 3256
🔢 EC Number — 202-396-2
⚖️ Molecular Weight — 135.19 g/mol
🔬 Molecular Formula — C₇H₅NS
📝 Odor Type — Sulfurous (with animalic, rubbery and meaty facets)
📈 Odor Strength — Extremely high. One of the most powerful materials on the perfumer's palette, in the same class of impact as pyrazines and dimethyl sulfide. Use levels typically in the 0.001–0.1% range in the final compound.
👃🏼 Odor Profile — Sulfurous, rubbery-latex, meaty, cooked, nutty, coffee-like, vegetable, quinoline-like; in extreme dilution reveals a ripe-fruity and slightly floral nuance.
⚗️ Uses — Flavor and fragrance agent; also a chemical intermediate for pharmaceuticals, dyes, optical brighteners and rubber vulcanization accelerators (via its 2-substituted derivatives).
🧴 Appearance — Clear yellow to yellow-brown, slightly viscous liquid.
Additional Physical Properties
🌡️ Melting Point — 2 °C (lit.)
🔥 Boiling Point — 227–233 °C at 760 mmHg (commonly reported as 231 °C lit.)
💧 Density — 1.238 g/mL at 25 °C (lit.)
🔍 Refractive Index — n²⁰/D 1.639–1.645 (lit. 1.642)
💧 Solubility — Slightly soluble in water (~3–4.3 g/L at 25 °C); freely soluble in ethanol, DPG, acetone, carbon disulfide and most common perfumery solvents and fixatives.
🔄 Specific Rotation — Not applicable (achiral, planar aromatic heterocycle).
✅ Purity — Commercial F&F grade typically ≥96% (GC); reagent grades up to ≥99%.
🧬 Biogenic Carbon Content — Typically 0% (synthetic, petrochemical origin). Bio-based grades are not routinely offered.
⚙️ Other — Vapor pressure 0.014 mmHg at 25 °C; vapor density 4.66 (air = 1); flash point > 110 °C (TCC); logP (o/w) ≈ 2.01; pKa ≈ 0.85 (predicted, conjugate acid).
What is Benzothiazole?
Benzothiazole is an aromatic bicyclic heterocycle formed by the ortho-fusion of a benzene ring with a 1,3-thiazole ring, giving the molecular formula C₇H₅NS. It is the parent compound of a very large family of industrially and biologically important derivatives (2-mercaptobenzothiazole, 2-aminobenzothiazole, firefly luciferin, riluzole, pramipexole, many dyes and vulcanization accelerators).
In nature, benzothiazole occurs at trace levels in a surprisingly wide range of foods and beverages – cranberries, coffee, black and green tea, cooked meat, boiled milk, tomato, popcorn, roasted cocoa, wine and many fermented products – where it contributes to cooked, roasted and sulfurous top-notes. In the environment, significant non-biogenic levels are associated with tire-wear particles, street run-off and rubber manufacturing, because 2-mercaptobenzothiazole and related vulcanization accelerators slowly release benzothiazole on weathering.
For perfumery and flavor use, benzothiazole is produced synthetically, most commonly by cyclization of 2-aminothiophenol with formic acid or an equivalent C1 donor, or by oxidative cyclization of N-arylthioamides.
Historical Background
The benzothiazole scaffold was first put on the chemical map by August Wilhelm von Hofmann in 1879, who reported the synthesis of 2-chloro- and 2-phenylbenzothiazoles and laid the groundwork for the classical Hofmann / Hugerschoff cyclization routes still used today.
Industrial interest exploded after 1921, when 2-mercaptobenzothiazole (MBT) and its disulfide were introduced as rubber vulcanization accelerators, making the benzothiazole nucleus one of the cornerstones of 20th-century rubber technology.
In the flavor and fragrance field, benzothiazole became relevant once modern GC-MS analysis of natural volatiles revealed its ubiquity in food aromas. Parent benzothiazole was first isolated from a natural source in 1967, from the volatiles of American cranberries (Vaccinium macrocarpon Ait., cv. Early Black). It was subsequently reviewed as a flavor ingredient by FEMA (GRAS #3256) and by JECFA (No. 1040, evaluated in 2002), and entered the perfumer's palette as a very-low-dosage high-impact material, documented by Arctander and later by Pellwall, The Good Scents Company and Perfumer & Flavorist.
Olfactory Profile
Scent Family
Sulfurous / animalic-synthetic, with rubbery-leathery and cooked-meaty facets and, in extreme dilution, a ripe-fruity and quinoline-like nuance.
Main Descriptors
Characteristic notes reported across technical sources: sulfurous, rubbery, latex, cooked, meaty, nutty, coffee-like, vegetable, quinoline-like, medicinal, cold, slightly phenolic. At > 0.1% it is unmistakably industrial (new tires, vulcanized rubber, shoemaker's workshop); at 1–10 ppm in a blend it reads as "boiled", "ripe" and "natural".
Intensity
Extremely high. Detection threshold in water is reported in the low-ppb range (≈ 1–10 μg/L). Perfumery use levels are typically 0.001–0.1% of the concentrate; flavor use levels (FEMA) are in the low-ppm range in the finished beverage or food.
Tenacity
Medium to long on the blotter (several hours to a day); excellent substantivity on skin and fabric thanks to the aromatic heterocyclic core and low vapor pressure.
Volatility
Heart-to-base note. Behaves as a heart note when heavily diluted in fruity or floral compositions, and as a base-note modifier in leather, animalic and industrial accords.
Applications in Fine Fragrance
Benzothiazole is essentially a seasoning molecule: it is almost never a dominant note, but it lifts and "humanises" a remarkable range of accords when used at trace level.
Fruity & fruity-floral accords (1–50 ppm of the concentrate) — Adds an authentic ripeness and a sulfurous "skin of the fruit" realism that cannot be achieved with esters and lactones alone. Particularly effective in blackcurrant, tropical, passion-fruit, guava and osmanthus-peach accords.
White florals (1–20 ppm) — Reinforces the natural indolic-animalic halo of jasmine, tuberose, gardenia and frangipani; reads as "living flower" rather than stylized bouquet.
Leather & rubber accords (10–500 ppm) — Primary workhorse for modern "industrial" leather, latex, neoprene and asphalt notes; pairs with isobutyl quinoline, birch tar, styrax and safraleine.
Animalic & skin bases (1–100 ppm) — Adds a sweaty, warm-body, quinoline-like facet to musk/ambrox bases, useful in skin-scent compositions.
Gourmand & coffee accords (1–20 ppm) — Supports roasted, boiled-milk, condensed-milk and coffee aspects together with pyrazines, furaneol and maltol.
Fantasy/industrial accords (50–1000 ppm) — Signature component for "new car", "sneaker", "hospital" and "plastic" olfactory exercises.
Performance in Formula
Fixative Power — Good; acts as a mild sulfurous fixative for fruity and floral notes.
Radiance Effect — Adds diffusion and "lift" to heavy bases, similar in function (though not in smell) to pyrazines.
Stability — Chemically stable under normal formula conditions; recommended storage under inert atmosphere, cool and dark, as the neat liquid is air-sensitive and can darken over time.
Compatibility — Compatible with the vast majority of perfumery ingredients; discolors only slightly in alcoholic solutions. Avoid strong oxidizers.
Solubility — Fully soluble in ethanol, DPG, IPM, TEC and most common fixative solvents at working dilution. Practically insoluble in water as neat material.
Industrial & Technical Uses
Functional Perfumery — Low-level modifier in fabric care, air care and household products where a "freshly-washed" or "clean rubber" nuance is desired.
Personal Care — Used in fine fragrance and eaux de toilette concentrates that are then incorporated into personal care products; not used as a standalone cosmetic active.
Flavor Industry — FEMA GRAS #3256; JECFA #1040. Applied at ppm-level in dairy (condensed milk, yogurt, cheese), coffee, cocoa, meat, savory, popcorn, nut, berry and tropical fruit flavors.
Cosmetics — Listed in the EU CosIng database with the function Fragrance; used exclusively as part of fragrance compounds, subject to IFRA/RIFM safe-use levels.
Non-F&F uses — Intermediate for optical brighteners, disperse and sensitizing dyes, photographic chemicals, corrosion inhibitors, pharmaceutical APIs (e.g. riluzole, pramipexole precursors) and, through its 2-mercapto derivative, rubber vulcanization accelerators.
Regulatory & Safety Overview
IFRA Status
Benzothiazole is listed on the IFRA Transparency List as a recognized fragrance ingredient. As of the 51st Amendment there is no standalone IFRA Standard that restricts or prohibits benzothiazole; safe use is governed by the RIFM Expert Panel's Quantitative Risk Assessment (see RIFM 2018 below). Perfumers should still respect the category-specific NESIL/AEL derived from that assessment.
EU Cosmetics Regulation
Permitted for use as a fragrance component under Regulation (EC) No 1223/2009. Not listed among the 24/26 declarable allergens of Annex III; not currently listed as a CMR or prohibited substance in Annex II. INCI name: Benzothiazole.
EU REACH
Fully registered under REACH (EC 202-396-2, CAS 95-16-9); current tonnage band, registrant list and dossier status should be verified directly on the ECHA CHEM substance factsheet before publication. Also listed in the CLP Inventory (notified classification).
FEMA Status
GRAS — FEMA #3256, published in the 5th GRAS list. Reaffirmed in later FEMA reviews; no longer subject to open safety questions for its declared flavor uses.
JECFA / Codex
JECFA flavor No. 1040, evaluated at the 59th meeting (2002). Conclusion: "no safety concern at current levels of intake when used as a flavouring agent". ADI: acceptable.
US FDA
Listed in the "Substances Added to Food" inventory (formerly EAFUS) as a flavoring agent / adjuvant (FEMA #3256). No separate FDA food additive regulation.
GHS / CLP Classification (notified, as per reference SDS)
Values below reflect the classification notified by major suppliers (e.g. TCI SDS B0092, Synerzine T720). Individual registrants may differ — always rely on the SDS of the exact lot/supplier in use.
H301 – Toxic if swallowed
H311 – Toxic in contact with skin
H319 – Causes serious eye irritation
H332 – Harmful if inhaled
H373 – May cause damage to organs (blood / blood system) through prolonged or repeated exposure
H402 – Harmful to aquatic life
Signal word: Danger. Pictograms: GHS06 (toxic), GHS07 (harmful), GHS08 (health hazard). These values refer to the neat material; standard 1–10% dilutions used in perfumery carry a much lower hazard profile.
Toxicology (summary from RIFM 2018 / Api et al.)
Skin Sensitization — Not a skin sensitizer at current use levels; QRA supports safe use within the derived AELs.
Phototoxicity / Photoallergy — Not phototoxic / not photoallergenic (UV/VIS spectrum does not meet criteria of concern).
Genotoxicity — Negative in Ames (OECD TG 471) and in BlueScreen assays, with and without metabolic activation.
Repeated Dose Toxicity — NOAEL reported at 5.1 mg/kg bw/day (read-across + target studies); MOE > 100 at current exposure estimates.
Reproductive / Developmental — No concerns identified at current use levels.
Local Respiratory Toxicity — No concerns at current use levels.
Environmental Considerations
Biodegradability — Not readily biodegradable; considered persistent in the environment, especially in aquatic and sediment compartments.
Bioaccumulation — Low bioaccumulation potential (logP ≈ 2).
Aquatic Toxicity — Harmful to aquatic life (H402). Benzothiazoles are classified as contaminants of emerging concern in urban run-off.
Sustainability Credentials
No mainstream renewable/biotech route is commercially offered for benzothiazole itself at the time of writing. Typical supply is petrochemical, with REACH-registered manufacture in the EU and Asia. No ISO 16128 natural index, no COSMOS/NATRUE approval and no ASTM D6866 bio-based carbon certification are available for the parent molecule.
Related Ingredients on Scentspiracy
Complementary materials in leather / rubber accords:
Birch Tar — reinforces the smoky-leather facet
Styrax — smooths the rubbery edge with balsamic warmth
Safraleine — adds a spicy-leather radiance
Isobutyl Quinoline — core leathery pairing
Complementary materials in fruity-floral accords:
Beta-Damascenone — amplifies the ripe-fruit effect
γ-Undecalactone — creamy peachy body
Ethyl Maltol — sweet gourmand lift
Osmanthus absolute / Tuberose absolute — natural indolic-fruity host
References
PubChem, CID 7222 – Benzothiazole. National Library of Medicine (NIH). https://pubchem.ncbi.nlm.nih.gov/compound/7222
NIST WebBook – Benzothiazole (CAS 95-16-9). National Institute of Standards and Technology. https://webbook.nist.gov/
Sigma-Aldrich / Merck KGaA, Product 101338 – Benzothiazole, ≥96%. Technical datasheet and SDS.
Tokyo Chemical Industry (TCI), Product B0092 – Benzothiazole. Product specifications and SDS.
The Good Scents Company – Benzothiazole, 95-16-9. http://www.thegoodscentscompany.com/data/rw1007521.html
Arctander, S. Perfume and Flavor Chemicals (Aroma Chemicals), 1969 – monograph on Benzothiazole.
Wright, J. "Flavor Bites: Benzothiazole." Perfumer & Flavorist, December 2021. https://www.perfumerflavorist.com/flavor/ingredients/article/21927822/
Api, A.M., Belsito, D., Botelho, D., et al. "RIFM fragrance ingredient safety assessment, benzothiazole, CAS Registry Number 95-16-9." Food and Chemical Toxicology 122 (Suppl. 1): S355–S361, 2018. doi:10.1016/j.fct.2018.09.030
Api, A.M., Bartlett, A., Belsito, D., et al. "Update to RIFM fragrance ingredient safety assessment, benzothiazole, CAS Registry Number 95-16-9." Food and Chemical Toxicology, 2024. doi:10.1016/j.fct.2024.115156
IFRA – Transparency List and IFRA Standards Library, 51st Amendment (2023). https://ifrafragrance.org/
FEMA (Flavor and Extract Manufacturers Association) – GRAS monograph 3256, Benzothiazole. https://www.femaflavor.org/flavor-library/benzothiazole
JECFA – Specifications for Flavourings, Benzothiazole, No. 1040, 59th meeting (2002). FAO/WHO. https://www.fao.org/food/food-safety-quality/scientific-advice/jecfa/jecfa-flav/details/en/c/973/
US FDA – Substances Added to Food (formerly EAFUS) – Benzothiazole.
ECHA – Substance Information, Benzothiazole, EC 202-396-2 / CAS 95-16-9. European Chemicals Agency. https://echa.europa.eu/ and https://chem.echa.europa.eu/100.002.179/
NTP – Nomination Background: Benzothiazole (CASRN 95-16-9). National Toxicology Program, NIEHS.
De Wever, H.; Verachtert, H. "Biodegradation and toxicity of benzothiazoles." Water Research 31(11): 2673–2684, 1997.
ChemicalBook – Benzothiazole (CAS 95-16-9), product properties. https://www.chemicalbook.com/ChemicalProductProperty_EN_CB1720657.htm
Wikipedia – Benzothiazole. https://en.wikipedia.org/wiki/Benzothiazole