Terpineol (α-Terpineol) - Technical Overview
🏭 Manufacturer — Industrial synthesis from α-pinene/turpentine; multiple global producers
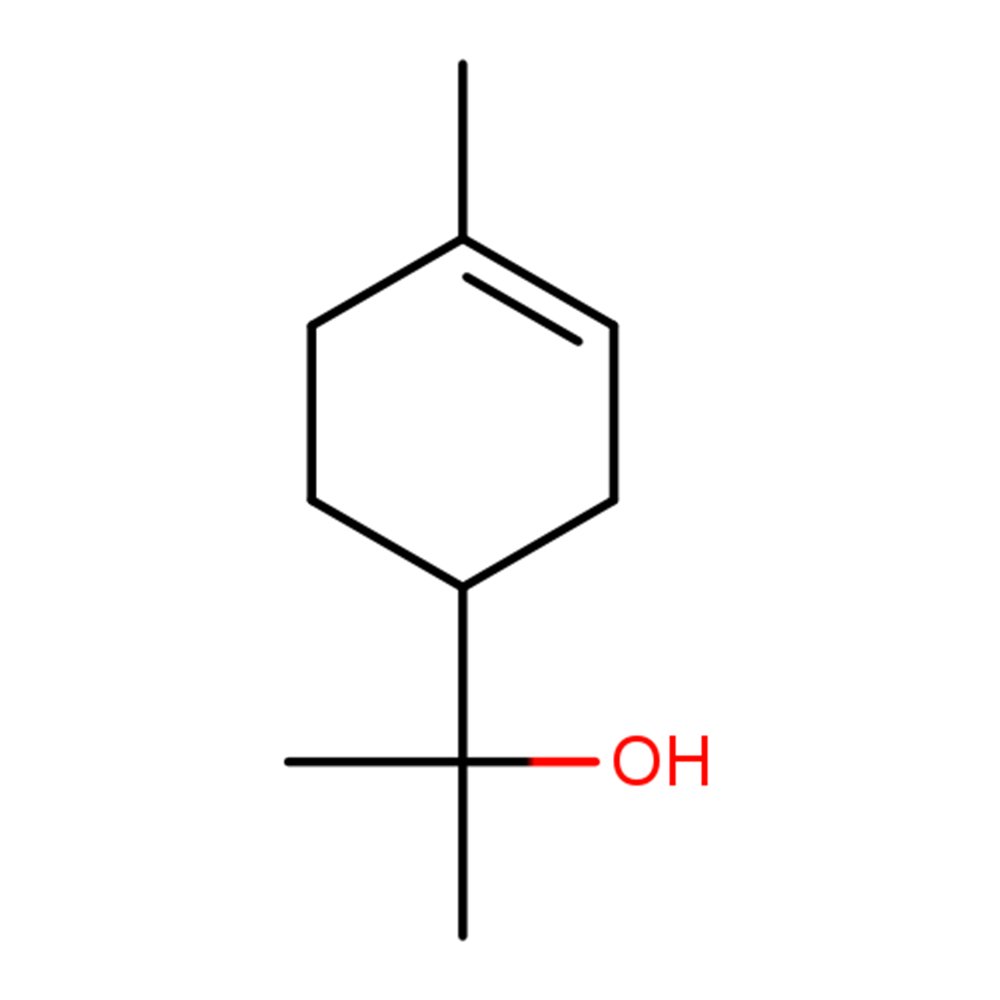
🔎 IUPAC Name — 2-(4-Methyl-1-cyclohex-3-enyl)propan-2-ol
🧪 Synonyms — α-Terpineol, β-Terpineol, γ-Terpineol, p-Menth-1-en-8-ol
🧬 Chemical Formula — C₁₀H₁₈O
📂 CAS Number — 98-55-5 (α-terpineol), 138-87-4 (β-terpineol), 586-81-2 (γ-terpineol), 8000-41-7 (mixed isomers)
📘 FEMA Number — 3049 (GRAS status)
⚖️ Molecular Weight — 154.25 g/mol
📝 Odor Type — Fresh-terpenic, floral (lilac)
📈 Odor Strength — Medium (approximately 20 hours on strip)
👃🏼 Odor Profile — Clean, fresh lilac-floral with terpenic brightness; citrus-soapy character with slight dirty undertone; mild peach nuance in drydown
⚗️ Uses — Terpenic modifier, floral booster, pine/fougère core component, household/functional perfumery, flavoring (citrus, coffee, tomato, apple accords)
🧴 Appearance — Colorless to pale yellow liquid (α-isomer)
♨️ Impact — Top-to-heart note with moderate persistence
What is Terpineol?
Terpineol refers to a family of cyclic monoterpene alcohols comprising four isomeric forms: α-terpineol (the most commercially important), β-terpineol, γ-terpineol, and terpinen-4-ol. The α-isomer, which accounts for the majority of commercial production, is the focus of this overview.
These alcohols occur naturally in numerous essential oils—including pine, petitgrain, cajuput, cardamom, clary sage, lavender, and eucalyptus—but at concentrations far too low to meet industrial demand (Arctander, 1960). Most commercial terpineol is semi-synthetic, derived from the acid-catalyzed hydration of α-pinene, a major constituent of turpentine oil obtained as a byproduct of the pine pulp and paper industry.
The α-isomer features a distinctive lilac-like floral scent with terpenic freshness, making it invaluable in both fragrance and flavor applications. It serves as a clean floral top-to-heart note, bridging citrus openings to green or floral middle notes in perfume compositions.
Historical Background
Early Terpene Chemistry & Structure Elucidation (1850s-1890s)
The story of terpineol begins with the broader investigation of terpene chemistry in the mid-19th century. The study of terpenoids occupied the attention of eminent organic chemists including Kekulé, Perkin, and Wallach since the early 1800s (ScienceDirect, 2021). Prior to modern analytical tools, monoterpenes from turpentine were the focus of much early research due to their relative structural simplicity compared to higher terpenes.
German chemist Otto Wallach (1847-1931, Nobel Prize 1910) made fundamental contributions to terpene chemistry through his investigations of turpentine pyrolysis products beginning in the 1880s. Wallach established the "isoprene rule," observing that terpenoids contain multiple isoprene units—a principle later mechanistically explained by Ruzicka in the 1920s-1930s.
The structure of terpineol was definitively determined in 1885-1895 by Otto Wallach, Ferdinand Tiemann, and Friedrich Wilhelm Semmler through systematic chemical degradation studies (1911 Encyclopædia Britannica; Pybus & Sell, 2018). Semmler, in 1895, obtained active (optically active) solid terpineol by replacing halogen atoms in the active monohydrobromide of limonene with hydroxyl groups, demonstrating the molecule's structural relationship to other monoterpenes.
Industrial Development & Commercial Synthesis (1900s-1930s)
By the early 1900s, the commercial importance of terpineol became evident as the fragrance industry sought cost-effective synthetic alternatives to expensive natural floral extracts. Terpineol emerged as a key ingredient for creating lilac-type fragrances—one of the first successfully reconstructed floral notes using synthetic materials (Arctander, 1960).
The industrial synthesis of terpineol developed through several key innovations:
1922: Robert Marchand (US Patent 1,408,462) used terpin hydrate as starting material, hydrolyzed under organic sulfonic acid catalysis to produce terpineol.
1930: Smith Lee T. and colleagues (US Patent 1,743,403) isolated α-terpineol directly from pine oil through fractional distillation, establishing the U.S. route of production from natural pine oil byproducts.
1930s-1940s: Multiple process improvements by Sheffield Donald H. (US Patents 2,060,597; 2,178,349; 2,336,575), Christian Meuly Walter (US Patent 2,088,030), and others refined both isolation and synthetic routes.
The Two-Step Method & Regional Production Differences
By mid-20th century, two distinct production philosophies emerged:
United States Approach: Terpineol produced mainly as an isolate from pine oil through fractional distillation. Pine oil, obtained from steam-distilled wood turpentine, naturally contains terpineol along with other monoterpene alcohols (Arctander, 1960).
European Approach: Terpineol considered a synthetic material, derived from turpentine oil through a two-step process:
Hydration of α-pinene (isolated from turpentine) with aqueous mineral acids to form crystalline cis-terpin hydrate
Partial dehydration of terpin hydrate with weak acids or acid-activated silica gel to yield α-terpineol plus isomers
This regional difference reflected feedstock availability: American turpentine contained 25-35% β-pinene (valuable for other products) and only ~50% α-pinene, making direct pine oil fractionation more economical. European and East Indian turpentines were rich in α-pinene but low in β-pinene, making synthetic conversion from α-pinene the preferred route (Arctander, 1960).
Golden Age of Soap Perfumery (1910s-1950s)
Terpineol achieved prominence as one of the most important fragrance ingredients for soaps from the 1910s well into the 1950s (David, 2023). In an era when soap perfumery represented mass-market fragrance, terpineol's combination of affordability, stability to alkali, and pleasant lilac character made it indispensable.
Early 20th-century soaps featured clear olfactory directions limited to affordable, chemically stable ingredients: bitter almond (benzaldehyde), rose (phenylethyl alcohol), violet (ionones), heliotrope (piperonal), lilac (terpineol), and carnation (eugenol). Terpineol's chemical stability in alkaline soap formulations—where many delicate naturals would decompose—secured its position as a workhorse ingredient (David, 2023).
Modern Production & Applications (1950s-Present)
Post-WWII systematic studies during the 1950s explored the olfactory diversity offered by synthetic molecules prepared from abundant terpenes using well-controlled processes. Terpene chemistry benefited from mechanistic understanding developed in the late 1940s-1950s regarding biosynthesis and cationic cyclization reactions (David, 2023).
Today, terpineol production relies primarily on crude sulfate turpentine (CST), a byproduct from the Kraft paper pulping process. Global turpentine production reaches approximately 300,000 metric tons annually, with α- and β-pinene (present in 7:3 ratio) serving as large-volume, low-cost starting materials for numerous fragrance substances (ScienceDirect, 2021).
Modern synthesis involves either:
Direct hydration of α-pinene or turpentine with acid catalysts (one-step method)
Hydration to terpin hydrate followed by selective dehydration (two-step method)
Biotransformation routes using microorganisms (emerging sustainable methods)
Terpineol remains valued for its stability, affordability, and versatility across fine fragrance, functional perfumery, and flavor applications.
Olfactory Profile
Scent Family
Fresh-Terpenic / Floral (Lilac), positioned within the floral-green category with clean, airy character
Main Descriptors
Primary: Lilac, fresh, clean, floral
Secondary: Terpenic, citrus-soapy, slightly peachy, Mild woody-balsamic undertone, faint "dirty" facet.
Intensity
Medium odor strength. Opens with terpenic citrus-clean impression with lemon-lime nuance, evolving to lilac floral-green character with faint soapy quality, finishing with light balsamic dryout retaining clean freshness. Moderate projection without overwhelming adjacent notes.
Longevity: Approximately 20 hours on blotter; a few hours of detectable presence on skin before retreating to allow heavier fixatives to dominate.
Tenacity
Moderate persistence. Functions as a bridge between volatile top notes and heavier base materials. Not a fixative but provides sufficient substantivity to carry through the heart phase of fragrance development.
Volatility
Medium-high volatility typical of monoterpene alcohols. Evaporates more slowly than terpene hydrocarbons (pinene, limonene) but faster than heavier alcohols (sandalwood derivatives). Appears within first minutes of evaporation and lingers long enough to bridge toward dry-down.
Boiling Point: 217-219°C at atmospheric pressure; 73.5°C at reduced pressure (0.5 kPa)
Fixative Role
Not a true fixative. Functions as a middle-note modifier that supports and extends floral character without significantly affecting overall composition longevity. May provide modest enhancement to perceived freshness duration in citrus-floral accords.
Applications in Fine Fragrance
Terpineol serves as a versatile floral modifier valued for its simplicity, affordability, and compatibility across multiple fragrance categories.
Core Applications
Lilac Reconstructions
Cornerstone ingredient in synthetic lilac accords (with heliotropin, cinnamic alcohol, anisalcohol)
Provides authentic lilac lift without heaviness
Combines with methyl anthranilate, cyclamen aldehyde, and phenylacetaldehyde derivatives
Floral Bouquets
Rounds rose accords without adding sweetness
Softens jasmine compositions that feel too indolic
Freshens muguet (lily-of-the-valley) accords that become overly sweet
Provides clean floral body in violet, narcissus, and hyacinth themes
Fougère & Pine Fragrances
Core component in masculine fougère structures
Supports coniferous sharpness in pine/forest accords
Bridges herbaceous lavender notes to woody bases
Combines effectively with oakmoss, coumarin, and lavender absolute
Citrus-Floral Transitions
Brightens citrus top notes while adding floral dimension
Softens sharp aldehydic openings
Creates seamless evolution from fresh opening to floral heart
Household & Functional Perfumery
Signature "clean" note in detergents, soaps, and air care
Stability to alkali makes it ideal for soap formulations
Cost-effective volume builder in budget-friendly compositions
Typical Usage Level: 0.5-5% in fine fragrance; 1-10% in functional perfumery and soap bases
Blending Partners
Florals: Linalool, phenylethyl alcohol, geraniol, nerol, rose absolute, jasmine, mimosa
Citrus: Lemon oil, d-limonene, bergamot, lime
Woody: Cedarwood derivatives, sandalwood, oakmoss
Aromatics: Lavender, lavandin, clary sage, isobornyl acetate, pine oil
Modifiers: Coumarin, ionones, methyl ionones, heliotropin
Performance in Formula
Blending Behavior
Terpineol demonstrates excellent compatibility with most perfume materials. Its medium polarity allows it to blend seamlessly with both polar (alcohols, aldehydes) and less polar (terpenes, musks) components.
Key Characteristics:
Pours cleanly and mixes without phase separation
Maintains clarity in alcohol-based formulations
Stable across typical perfumery pH ranges
Does not discolor or develop off-notes over time
Stability & Formulation
Alcohol Solubility: Excellent solubility in ethanol and dipropylene glycol (DPG)
Water Solubility: Limited; requires alcoholic carriers for aqueous systems
Alkali Stability: Excellent; one of few terpene alcohols stable in soap formulations
Oxidation Resistance: Moderate; less prone to oxidation than terpene hydrocarbons but benefits from antioxidant protection in long-term storage
Heat Stability: Stable at typical formulation temperatures; avoid prolonged heating above 80°C
Dosage Considerations
Low-Cost Volume Builder: Can be used at higher percentages (5-15%) in budget formulations to add floral body
Lilac Accent: 1-3% provides noticeable lilac character without dominating
Freshness Modifier: 0.5-1% adds clean, terpenic lift to complex accords
Formulation Tip: Start at 1-2% and adjust based on desired intensity; terpineol rarely overwhelms at normal usage levels, making it forgiving for beginner perfumers.
Industrial & Technical Uses
Food Flavoring (FEMA 3049)
GRAS Status: Approved for use as flavoring agent in food products
Applications:
Citrus Flavors: Enhances lemon, lime, and grapefruit profiles with terpenic freshness
Fruit Complexes: Apple, peach, pear, and tropical fruit flavors
Beverage Modifiers: Adds complexity to soft drinks and juice formulations
Coffee & Spice: Subtle use in coffee flavoring and tomato-based seasonings
Typical Levels: 1-10 ppm in finished food products
Functional Perfumery
Household Cleaners: Signature clean scent in multi-surface cleaners and detergents
Air Care: Provides fresh, clean notes in air fresheners and fabric sprays
Personal Care: Soaps, shampoos, body washes, and deodorants
Industrial Applications: Masking agents, institutional cleaning products
Pharmaceutical & Cosmetic
Antimicrobial Properties: Exhibits bactericidal and fungicidal activity; used in disinfectant formulations
Anti-inflammatory Effects: Topical applications in ointments and creams
Penetration Enhancer: Improves transdermal delivery of active ingredients
Oral Care: Occasional use in mouthwashes and toothpastes for fresh flavor
Emerging Applications
Recent research has identified additional biological properties including antioxidant, antiproliferative, and analgesic effects, suggesting potential applications beyond traditional fragrance and flavor uses (da Silva Rivas et al., 2020).
Regulatory & Safety Overview
IFRA Status
IFRA Amendment 51 (2023): Not restricted. No specific concentration limits across Categories 1-12.
EU Allergens
Not listed among the 26 declarable allergens under EU Cosmetics Regulation 1223/2009. However, presence should be verified in finished products as trace levels may trigger declaration thresholds in complex formulations.
FEMA Status
FEMA 3049 — GRAS (Generally Recognized As Safe) for use as flavoring ingredient
ECHA (REACH)
Registered substance under EU REACH regulations (CAS 98-55-5, 8000-41-7). Not classified as hazardous under standard perfumery and cosmetic use conditions.
Toxicology Summary
Oral LD₅₀ (rat): >2000 mg/kg (low acute toxicity)
Dermal Irritation: Minimal to no irritation at typical use concentrations
Sensitization: Not classified as a skin sensitizer
Phototoxicity: No evidence of phototoxic effects
Environmental: Biodegradable; moderate aquatic toxicity at high concentrations
Safety Note: Low irritancy under normal usage conditions. Some individuals may experience sensitivity; standard safety precautions recommended.
Handling Precautions
Use in well-ventilated areas
Avoid direct inhalation of concentrated vapors
Wear appropriate personal protective equipment (gloves, safety glasses)
Dilute before evaluation to prevent olfactory fatigue
Additional Technical Information
Production Methods
Chemical Synthesis Routes:
Terpin Hydrate Method (Two-Step, Traditional):
Hydration of α-pinene with aqueous mineral acids → cis-terpin hydrate (mp 117°C)
Partial dehydration with weak acids or acid-activated silica gel → α-terpineol + isomers
Higher purity but longer reaction time
Direct Hydration (One-Step, Modern):
Selective hydration of α-pinene, 3-carene, limonene, or turpentine without terpin hydrate formation
Addition of organic acids (with catalytic mineral acids) produces terpinyl esters
Subsequent hydrolysis (sometimes in situ) yields terpineol
Simpler process but may produce more byproducts (fenchol, borneol, cineol)
Pine Oil Isolation (U.S. Method):
Fractional distillation of yellow or white pine oil
Isolates naturally occurring terpineol along with other monoterpene alcohols
Economical for markets with abundant pine oil byproducts
Biotransformation (Emerging Sustainable Route):
Microbial conversion using Sphingobium sp., Penicillium digitatum, Cladosporium sp.
Biotransformation of limonene, α-pinene, or β-pinene
Produces optically active terpineol forms
Lower yields but environmentally preferable
Physical Properties
Density (d²⁰₄): 0.9315
Refractive Index (n²⁰ᴅ): 1.4799
Boiling Point (101.3 kPa): 212-219°C
Boiling Point (0.5 kPa): 73.5°C
Appearance: Colorless, slightly viscous liquid (pours more slowly than water)
Solubility: Good in ethanol, oils; limited in water
Isomer Composition
Commercial terpineol is typically a mixture of isomers:
α-Terpineol: 60-95% (main odorant)
γ-Terpineol, β-Terpineol: Minor components
Terpinen-4-ol: Byproduct from terpin hydrate method; contributes spicy, nutmeg-like, woody-earthy facets
Quality Control
GC analysis for isomer composition and purity
Organoleptic evaluation for characteristic lilac odor
Absence of off-notes from oxidation or contamination
Refractive index and density measurements for identity confirmatio
References
Arctander, S. (1960). Perfume and flavor materials of natural origin. Published by the author.
da Silva Rivas, A. C., Lopes, P. M., de Barros Azevedo, M. M., Duarte, D. M. C., & Sampai o, B. M. (2020). Production, properties, and applications of α-terpineol. Food and Bioprocess Technology, 13, 1261-1279. https://doi.org/10.1007/s11947-020-02461-6
David, F. (2023). Industrial fragrance chemistry: A brief historical perspective. European Journal of Organic Chemistry, e202300900. https://doi.org/10.1002/ejoc.202300900
Pybus, D. H., & Sell, C. S. (Eds.). (2018). The chemistry of fragrances: From perfumer to consumer (2nd ed.). Royal Society of Chemistry.
ScienceDirect. (2021). Terpineol - an overview. In Comprehensive Natural Products II. Elsevier. https://www.sciencedirect.com/topics/neuroscience/terpineol
Wallach, O., Tiemann, F., & Semmler, F. W. (1885-1895). Structure determination of terpineol. Berichte der Deutschen Chemischen Gesellschaft.
1911 Encyclopædia Britannica/Terpenes. (1911). Encyclopædia Britannica (11th ed.). https://en.wikisource.org/wiki/1911_Encyclopædia_Britannica/Terpenes
Patent References
Marchand, R. (1922). Process of making terpineol (US Patent 1,408,462). United States Patent Office.
Smith, L. T. et al. (1930). Isolation of α-terpineol from pine oil (US Patent 1,743,403). United States Patent Office.
Regulatory & Standards References
FEMA (Flavor and Extract Manufacturers Association). (n.d.). FEMA 3049 - Terpineol. FEMA GRAS Database.
IFRA (International Fragrance Association). (2023). IFRA Standards — 51st Amendment. https://ifrafragrance.org/